Recombinant Cynomolgus Monkey MMP-9 Protein, CF
R&D Systems, part of Bio-Techne | Catalog # 10833-MP

Key Product Details
- R&D Systems CHO-derived Recombinant Cynomolgus Monkey MMP-9 Protein (10833-MP)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
CHO
Accession #
Structure / Form
Proform
Conjugate
Unconjugated
Applications
Enzyme Activity
Product Specifications
Source
Chinese Hamster Ovary cell line, CHO-derived cynomolgus monkey MMP-9 protein
Ala20-Asp707
Ala20-Asp707
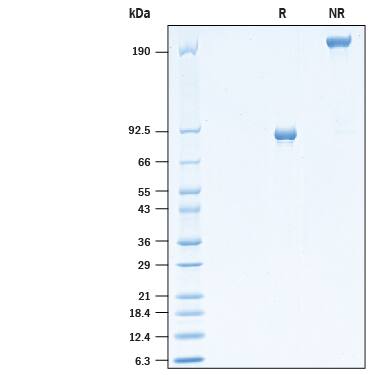
Purity
>90%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Ala20
Predicted Molecular Mass
76 kDa
SDS-PAGE
83-95 kDa, under reducing conditions
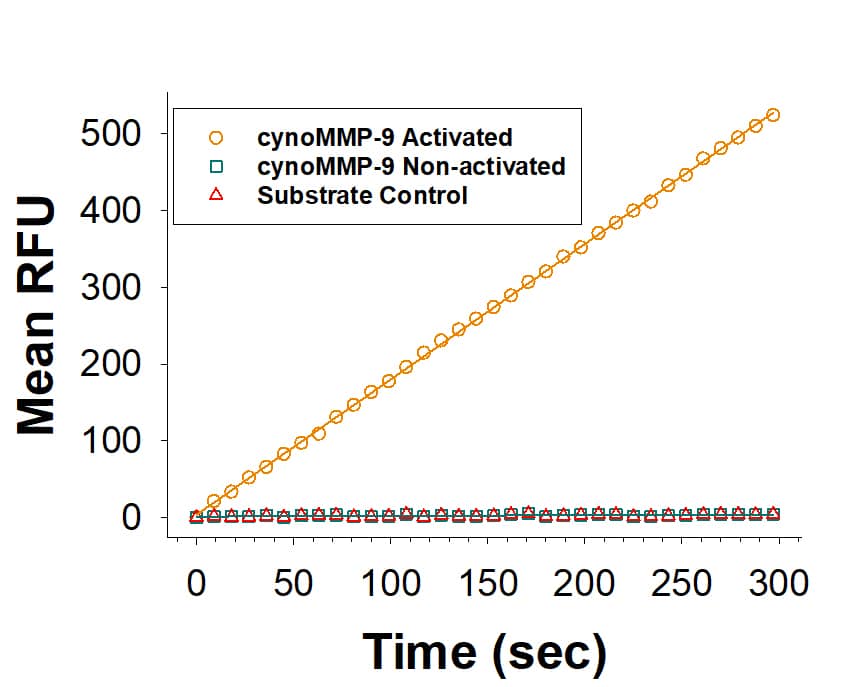
Activity
Measured by its ability to cleave the fluorogenic peptide substrate, Mca-PLGL-Dpa-AR-NH2 (Catalog # ES001).
The specific activity is >750 pmol/min/μg, as measured under the described conditions.
The specific activity is >750 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Cynomolgus Monkey MMP-9 Protein, CF
Recombinant Cynomolgus Monkey MMP-9 Enzyme Activity
Recombinant Cynomologous Monkey MMP-9 Protein (Catalog # 10833-MP) is measured by its ability to cleave the fluorogenic peptide substrate, Mca-PLGL-Dpa-AR-NH2 (ES001).Recombinant Cynomolgus Monkey MMP-9 Protein SDS-PAGE.
2 μg/lane of Recombinant Cynomolgus Monkey MMP-9 (Catalog # 10833-MP) was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing bands at ~90 kDa under reducing conditions.Formulation, Preparation and Storage
10833-MP
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, CaCl2, NaCl and Brij-35. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: MMP-9
References
- Kridel, S.J. et al. (2001) J. Biol. Chem. 276:20572.
- Vaisar, T. et al. (2009) Mol. Cell. Proteom. MCP 8:1044.
- Dufour, A. and C.M. Overall. (2013) Trends Pharmacol. Sci. 34:233.
- Vandooren, J. et al. (2013) Crit. Rev. Biochem. Mol. Biol. 48:222.
- Roeb, E. et al. (2002) J. Biol. Chem. 277:50326.
- Rosenblum, G. et al. (2007) Structure 15:1227.
- Kjeldsen, L. et al. (1993) J. Biol. Chem. 268:10425.
- Olson, M.W. et al. (2000) J. Biol. Chem. 275:2661.
- Huang, H. (2018) Sensors 18:3249.
- Ram, M. et al. (2006) J. Clin. Immunol. 26:299.
- Hu, J. et al. (2007) Nat. Rev. Drug. Discov. 6:480.
- Fields, G.B. (2019) Cells. 8:984.
- Kumar, G.B. et al. (2019) Medchemcomm. 10:2024.
Long Name
Matrix Metalloproteinase 9
Alternate Names
CLG4B, Gelatinase B, GELB, MANDP2, MMP9
Gene Symbol
MMP9
UniProt
Additional MMP-9 Products
Product Documents for Recombinant Cynomolgus Monkey MMP-9 Protein, CF
Product Specific Notices for Recombinant Cynomolgus Monkey MMP-9 Protein, CF
For research use only
Loading...
Loading...
Loading...