Human/Mouse/Rat FKBP12 Antibody
R&D Systems, part of Bio-Techne | Catalog # MAB3777

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Gly2-Glu108
Accession # P62942
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human/Mouse/Rat FKBP12 Antibody
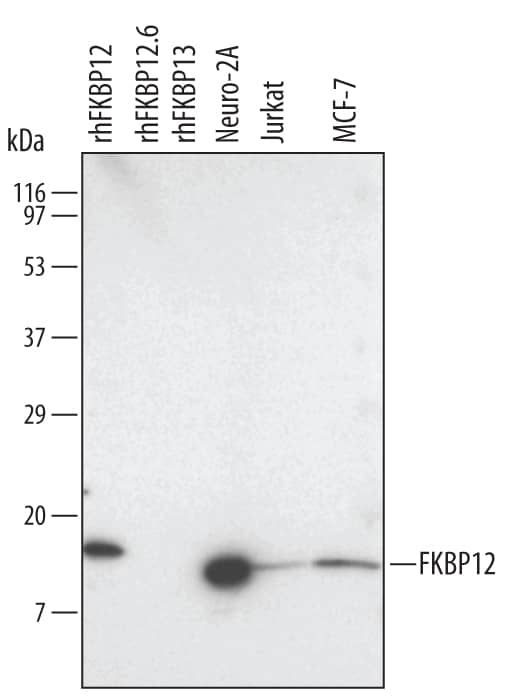
Detection of Human/Mouse/Rat FKBP12 by Western Blot.
Western blot shows lysates of Neuro-2A mouse neuroblastoma cell line, Jurkat human acute T cell leukemia cell line, and MCF-7 human breast cancer cell line. PVDF membrane was probed with 1 µg/mL Rat Anti-Human/Mouse/Rat FKBP12 Monoclonal Antibody (Catalog # MAB3777) followed by HRP-conjugated Anti-Rat IgG Secondary Antibody (Catalog # HAF005). For additional reference, Recombinant Human FKBP12 (Catalog # 3777-FK), recombinant human FKBP12.6, and recombinant human FKBP13 (5 ng/lane) were included. A specific band for FKBP12 was detected at approximately 12 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 3.Detection of Mouse FKBP12 by Immunocytochemistry/ Immunofluorescence
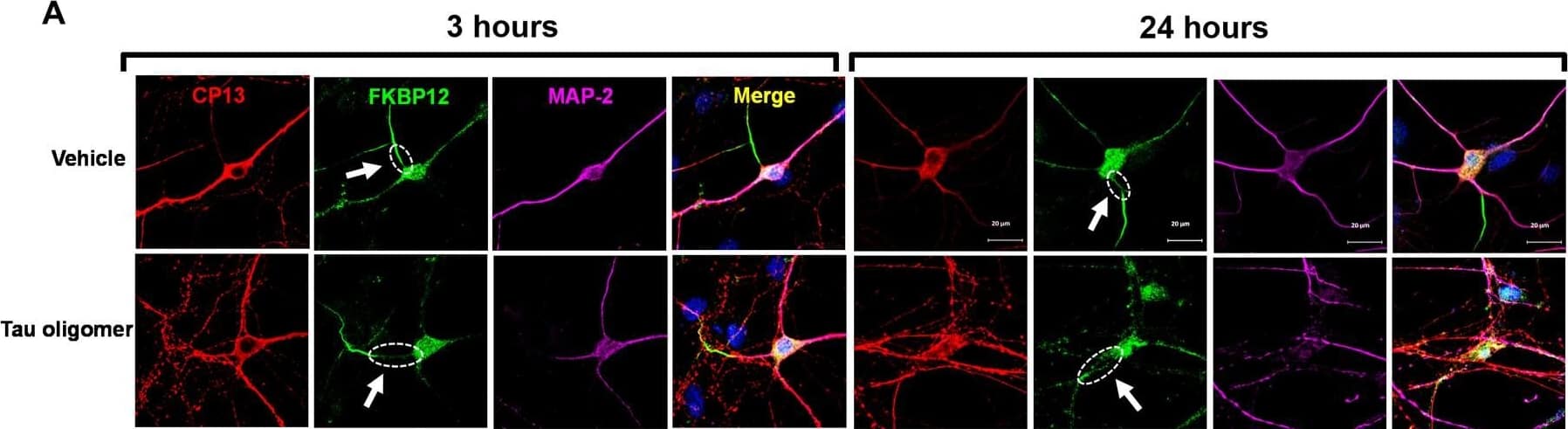
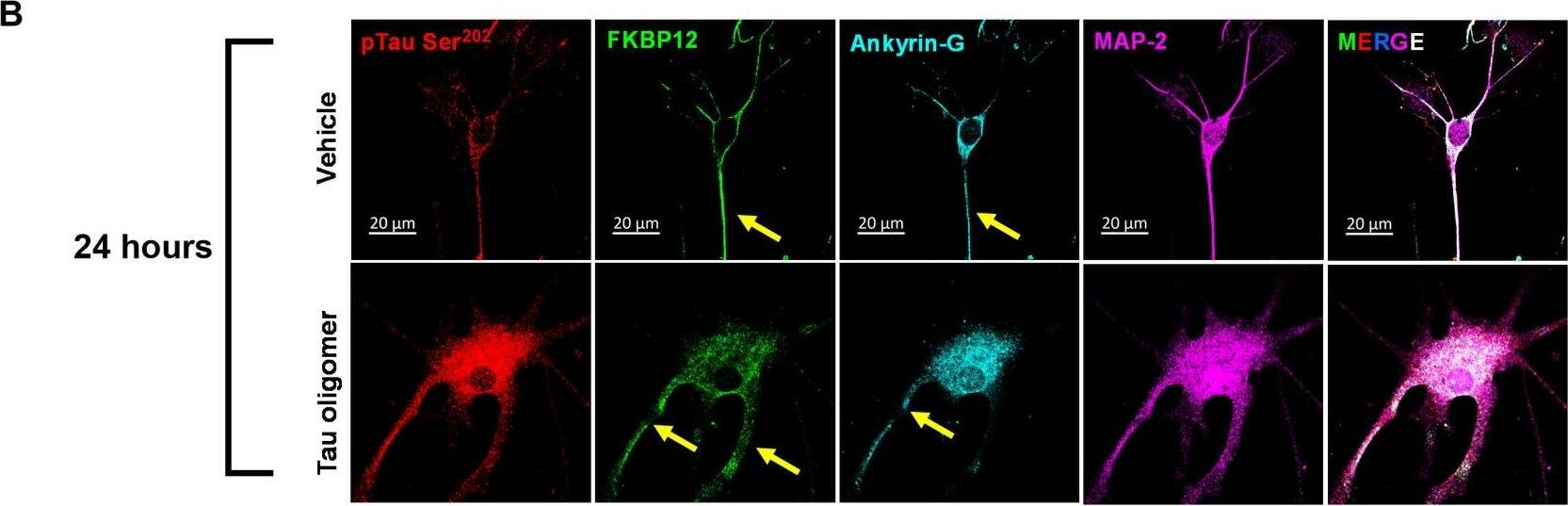
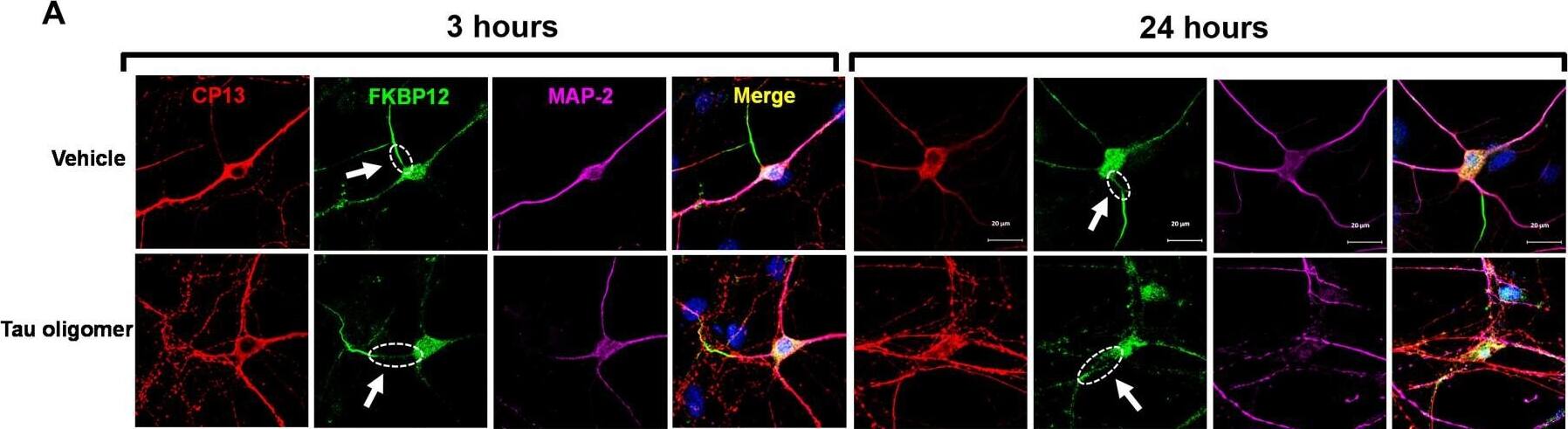
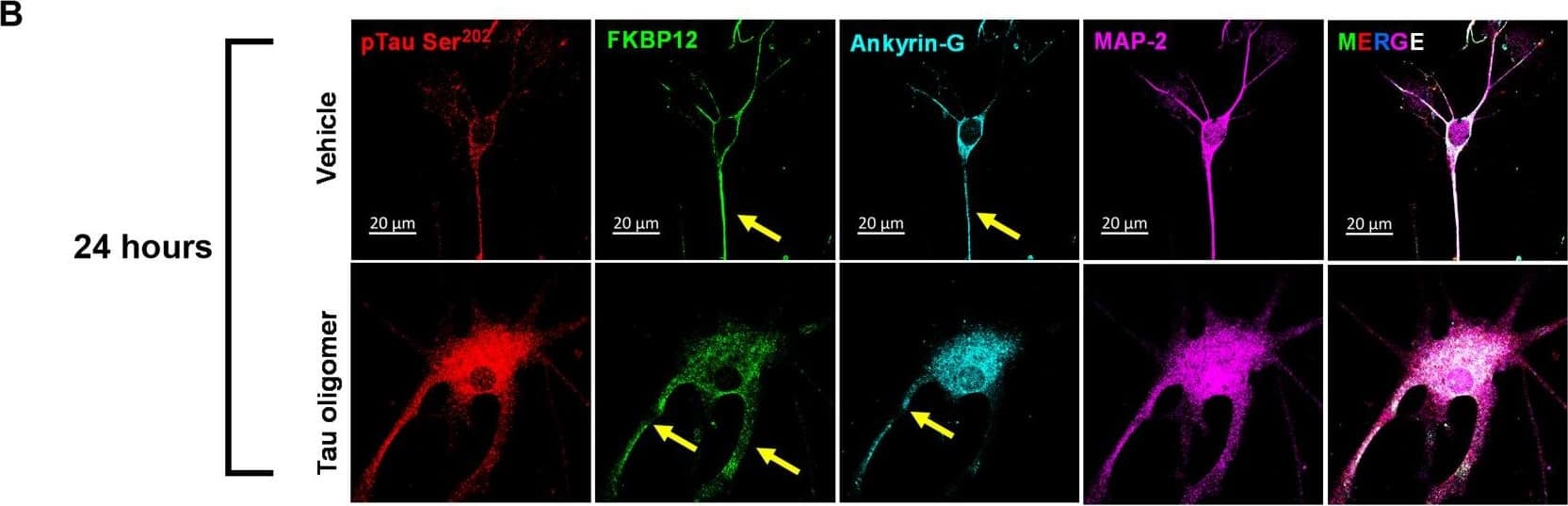
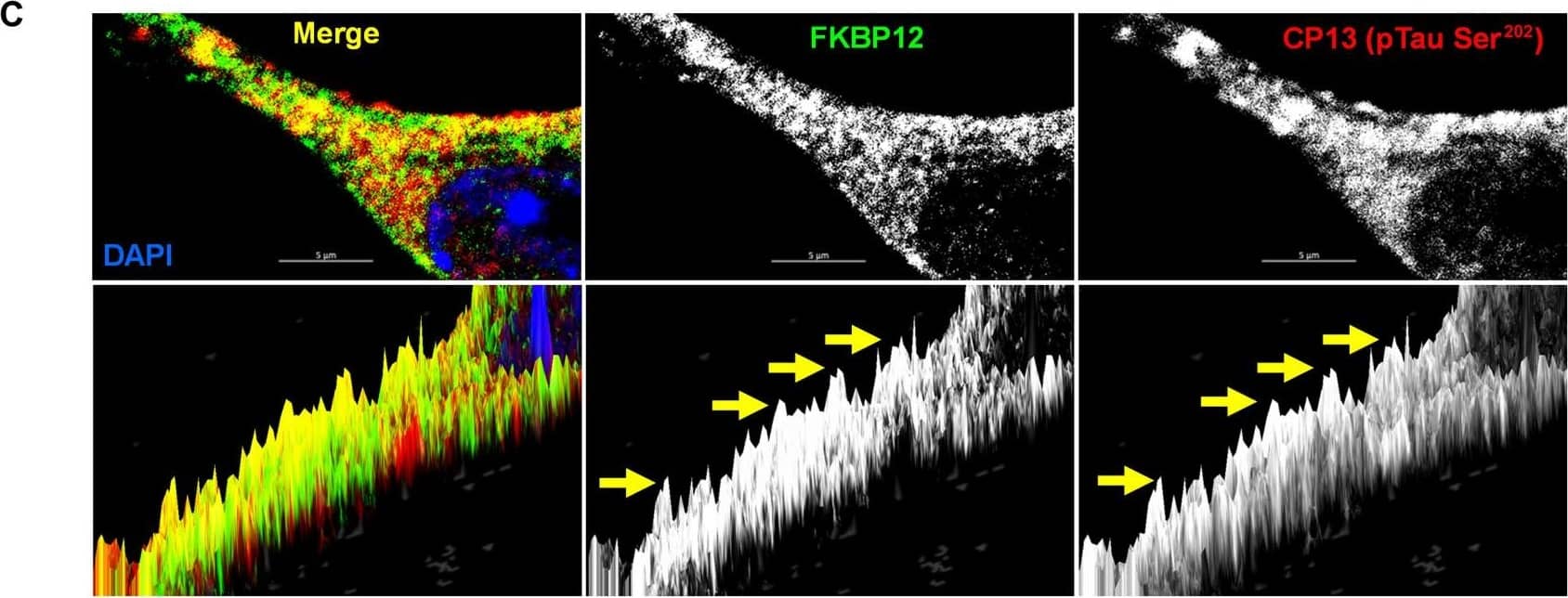
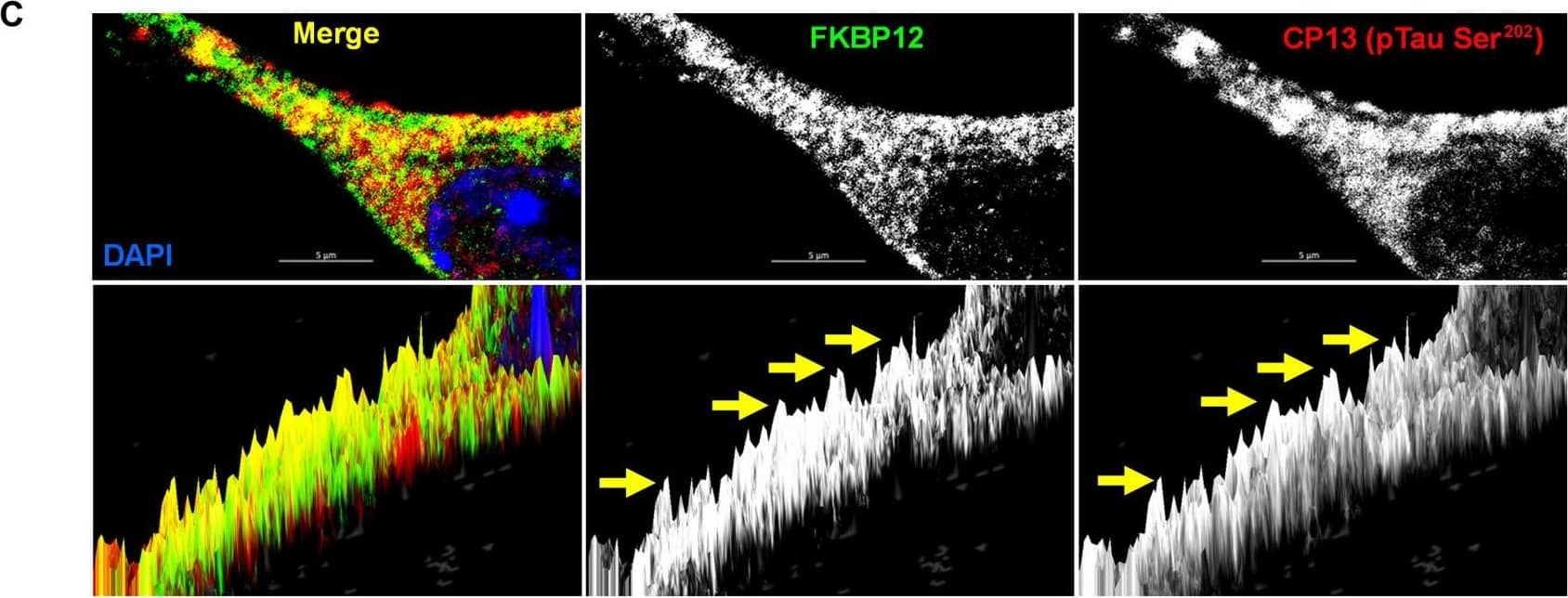
FKBP12 was translocated from axonal hillock to soma and colocalized to oTau.(A) Representative images of tau phosphorylation (CP13 antibody, red) and FKBP12 translocation in primary cortical neurons after induction of tau aggregation by oligomeric S1p fraction. Scale bars, 20 μm. (B) Representative images showed the high expression level of FKBP12 (green) in axonal hillock/axon initial segment (labeled by anti–ankyrin-G antibody, bright blue) under basal conditions whereas FKBP12 translocated to soma and dendrites when neurons bear tau aggregation. Scale bars, 20 μm. (C) Representative images showed the spatial colocalization of FKBP12 and aggregated Tau in the neurons after 24 hours of oTau seeding. Scale bars, 5 μm. DAPI, 4′,6-diamidino-2-phenylindole. (D) Quantification of FKBP12 intensity in the axon hillock of the neurons at 3 and 24 hours of S1p treatment, respectively. Data are expressed as means ± SEM. N = 10. Statistics by two-way ANOVA, post hoc multiple comparisons test by Fisher’s least significant difference (LSD). *P < 0.05, ***P < 0.005, and ****P < 0.001. (E and F) Quantification of granular intensity of CP13-labeled tau aggregates (E) and FKBP12 (F) in neurons at 3 and 24 hours of S1p treatment, respectively. Data are expressed as means ± SEM. N = 10. Statistics by two-way ANOVA, and post hoc multiple comparisons test by Fisher’s LSD. ****P < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36724228), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse FKBP12 by Immunohistochemistry
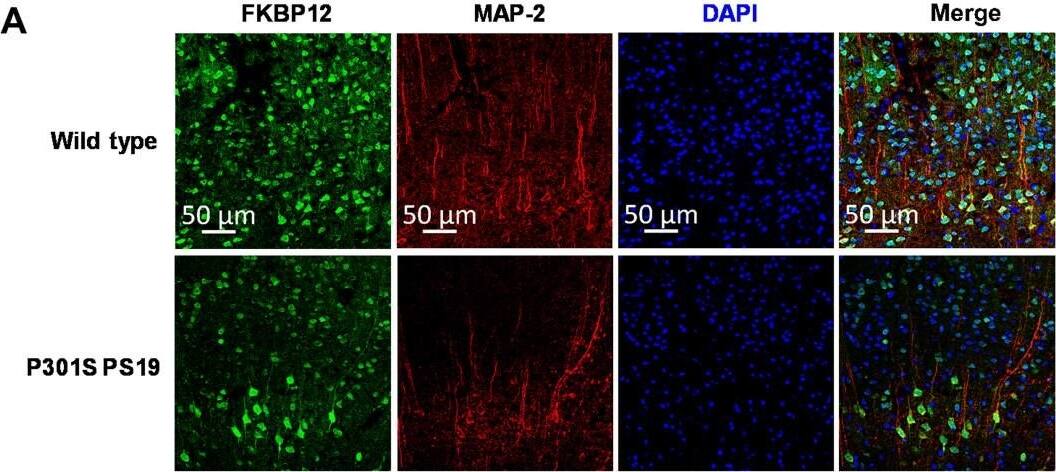
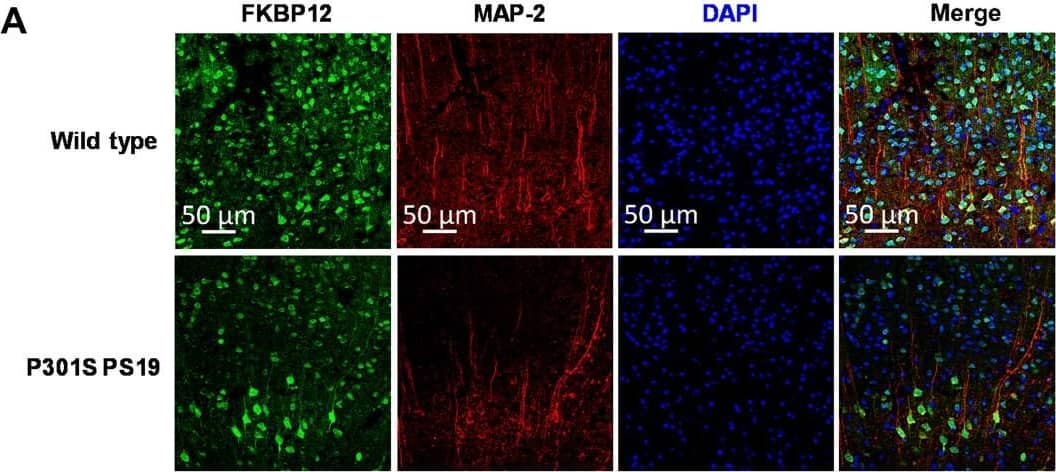
FKBP12 is decreased in PS19 mice brain.(A) Representative images showing the distribution of FKBP12 in entorhinal cortex of mouse brain. Scale bars, 50 μm. (B) Quantification of MAP-2 and FKBP12 fluorescence intensity in entorhinal cortex of PS19 mice brain in comparison to age-matched wild type (WT). Data are expressed as means ± SEM. N = 5. Statistics by unpaired t test, ***P < 0.005. (C) Representative Western blot showing high–molecular weight (HMW) tau aggregation in PS19 mice brain lysate and the decrease of FKBP12 in comparison to wild type. GAPDH, glyceraldehyde-3-phosphate dehydrogenase. (D) Quantification of the tau-5 band intensity showing the robust increase of the ratio between high–molecular weight tau to monomeric tau. (E) Quantification of the FKBP12 band intensity. (F) Representative Western blot images showing the expression level of phosphorylated tau (CP13 and PHF-1), FKBP12, and neuronal markers [postsynaptic density 95 (PSD-95) and MAP-2] in the brain lysate of 3-, 6-, and 9-month-old PS19 mice, respectively. (G) Quantification of the phosphorylated tau (CP13 and PHF-1) band intensity. Result was normalized by internal control of corresponding GAPDH band intensity. (H) Quantification of FKBP12 Western blot (WB) band intensity normalized by GAPDH and MAP-2, respectively. Statistics by one-way analysis of variance (ANOVA), post hoc multiple comparisons test by Tukey’s test. (I) Quantification for the band intensity of neuronal markers including PSD-95 and MAP-2, normalized by GAPDH. Data are expressed as means ± SEM. N = 3. Statistics by two-way ANOVA, and post hoc multiple comparisons test by Tukey’s. **P < 0.01, ***P < 0.005, and ****P < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36724228), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse/Rat FKBP12 Antibody
Western Blot
Sample: Neuro-2A mouse neuroblastoma cell line, Jurkat human acute T cell leukemia cell line, and MCF-7 human breast cancer cell line
Reviewed Applications
Read 1 review rated 5 using MAB3777 in the following applications:
Formulation, Preparation, and Storage
Purification
Reconstitution
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Background: FKBP12
FK506 binding protein, 12 kilodalton molecular weight (FKBP12), also called FKBP1, was originally characterized as a peptidyl-prolyl isomerase that catalyzes the transition between cis- and trans-proline residues critical for proper folding of proteins. Proline isomerase activity was demonstrated but not used for quality control. The macrolide immunosuppressants FK506 (Tacrolimus) and rapamycin bind to FKBP12 with high affinity, while the structurally related compound cyclosporine binds with a much lower affinity (1). The binding of these drugs causes FKBP12 to become a potent inhibitor of calcineurin phosphatase activity (2) and TOR kinase activity (3). The inhibition of protein phosphatase activity is highly selective for calcineurin (2), making the FK506/FKBP12 complex a useful tool in the study of this enzyme. Knockout mice lacking FKBP12 are morphologically normal, but develop cardiomyopathies that may be related to dysregulation of ryanodyne receptors (4).
References
- Hamilton, G.S. and J.P. Steiner (1998) J. Med. Chem. 41:5119.
- Liu, J. et al. (1992) Biochemistry 31:3896.
- Toral-Barza, L. et al. (2005) Biochem. Biophys. Res. Comm. 332:304.
- Hamilton , S.L and M.M. Matzuk (1998) Nature 391:489.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional FKBP12 Products
Product Documents for Human/Mouse/Rat FKBP12 Antibody
Product Specific Notices for Human/Mouse/Rat FKBP12 Antibody
For research use only