Recombinant Mouse Pentraxin 2/SAP His-tag Protein, CF
R&D Systems, part of Bio-Techne | Catalog # 2558-SAB

Key Product Details
- R&D Systems NS0-derived Recombinant Mouse Pentraxin 2/SAP His-tag Protein (2558-SAB)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Product Specifications
Source
Gln21-Asp224, with a C-terminal 10-His tag
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
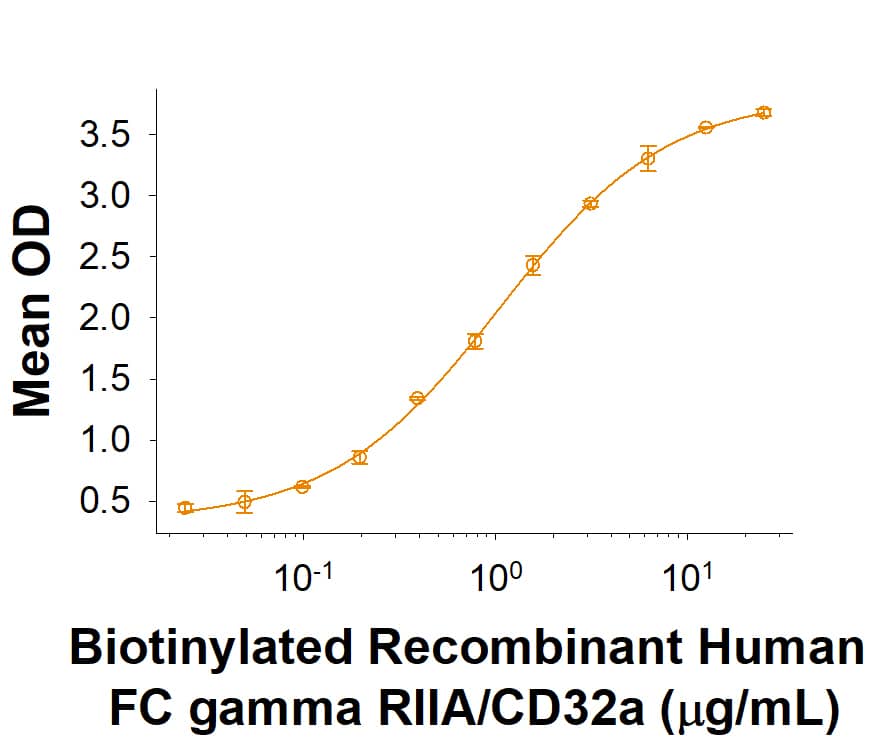
When recombinant Mouse Pentraxin/SAP His-tag (2558-SAB) is immobilized at 10 μg/mL (100 µL/well), the concentration of Biotinylated recombinant human FC gamma RIIA/CD32a (R167) Protein that produces 50% of the optimal binding response is found to be approximately 0.5-2.5 µg/mL.
Reviewed Applications
Read 1 review rated 5 using 2558-SAB in the following applications:
Scientific Data Images for Recombinant Mouse Pentraxin 2/SAP His-tag Protein, CF
Recombinant Mouse Pentraxin 2/SAP His-tag Protein Binding Activity
When Recombinant Mouse Pentraxin/SAP His-tag (2558-SAB) is immobilized at 10 μg/mL (100 μL/well), the concentration of Biotinylated Recombinant Human FC gamma RIIA/CD32a (R167) that produces 50% of the optimal binding response is found to be approximately 0.5-2.5 μg/mL.Formulation, Preparation and Storage
2558-SAB
| Formulation | Lyophilized from a 0.2 μm filtered solution in MOPS, NaCl and CaCl2. |
| Reconstitution |
Reconstitute at 100 μg/mL in sterile water.
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Pentraxin 2/SAP
Pentraxin 2 (also known as Serum Amyoid P Component or SAP) is a secreted glycoprotein that is a universal non-fibrillar component of amyloid deposits. Amyloid is an abnormal extracellular deposit of insoluble protein fibrils that can lead to tissue damage and disease (1-3). Pentraxin 2 belongs to the pentraxin (pentaxin) family, whose members have a characteristic pentagonal discoid arrangement of five non-covalently bound subunits (4). Pentraxin domains contain the consensus sequence, HxCx(S/T)WxS (x = any amino acid), a lectin fold, and two calcium-binding sites (1). They bind to a variety of unrelated molecules in a calcium-dependent lectin-like manner (1, 4, 5). Pentraxin 2 and C-reactive protein (CRP) are members of the classical or short pentraxin subfamily and share 46% amino acid (aa) identity (1). Mouse Pentraxin 2 is the major acute-phase protein whose expression is dependent on complement activation, IL-6 and/or IL-1 beta, while in humans, CRP is the major acute-phase protein (2, 5, 9). Both are produced and secreted by liver hepatocytes and circulate in plasma. The 204 aa mature mouse Pentraxin 2 shares 79% aa identity with rat Pentraxin 2 and 63-68% aa identity with human, guinea pig, golden hamster, porcine, and bovine Pentraxin 2 (2, 5). Amyloid deposits containing Pentraxin 2 are implicated in a diverse range of diseases including Alzheimer’s, prion diseases, type 2 diabetes and various systemic amyloidoses (3, 6, 7). Pentraxin 2 regulates the solubility of amyloid fibrils and protects them from degradation. In addition to its pathogenic role, Pentraxin 2 also has an important physiological function in innate immunity (8). It is an opsonin that interacts with all three types of human Fc gamma receptors that mediate neutrophil phagocytosis (8). Pentraxin 2 has been proposed to bind and sequester a variety of ligands including auto-antigens, apoptotic cells, chromatin, DNA, and micro-organisms (1-3). Pentraxin 2 is also a normal component of basement membranes (1).
References
- Garlanda, C. et al. (2005) Annu. Rev. Immunol. 23:337.
- Steel, D. and A. Whitehead (1994) Immunol. Today 15:81.
- Alexandrescu, A.T. (2005) Protein Sci. 14:1.
- Emsley, J. et al. (1994) Nature 367:338.
- Mole, J.E. et al. (1988) J. Immunol. 141:3642.
- Gillmore, J.D. et al. (2004) Immunology 112:255.
- Pepys, M. et al. (2002) Nature 417:254.
- Bharadwaj, D. et al. (2001) J. Immunol. 166:6735.
- Szalai, A.J. et al. (2000) J. Immunol. 165:1030.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional Pentraxin 2/SAP Products
Product Documents for Recombinant Mouse Pentraxin 2/SAP His-tag Protein, CF
Product Specific Notices for Recombinant Mouse Pentraxin 2/SAP His-tag Protein, CF
For research use only