Recombinant Human Muscle Glycogen Phosphorylase His-tag, CF
R&D Systems, part of Bio-Techne | Catalog # 11786-PM

Key Product Details
Product Specifications
Source
Human embryonic kidney cell, HEK293-derived human Glycogen phosphorylase, muscle form protein
Ser2-Ile842, with an N-terminal Met and 6-His tag
Ser2-Ile842, with an N-terminal Met and 6-His tag
Purity
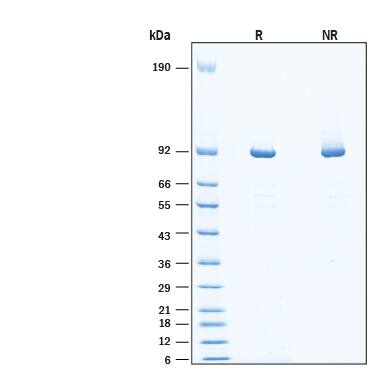
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Protein identity confirmed by mass spectrometry.
Predicted Molecular Mass
98 kDa
SDS-PAGE
86-96 kDa, under reducing conditions
Activity
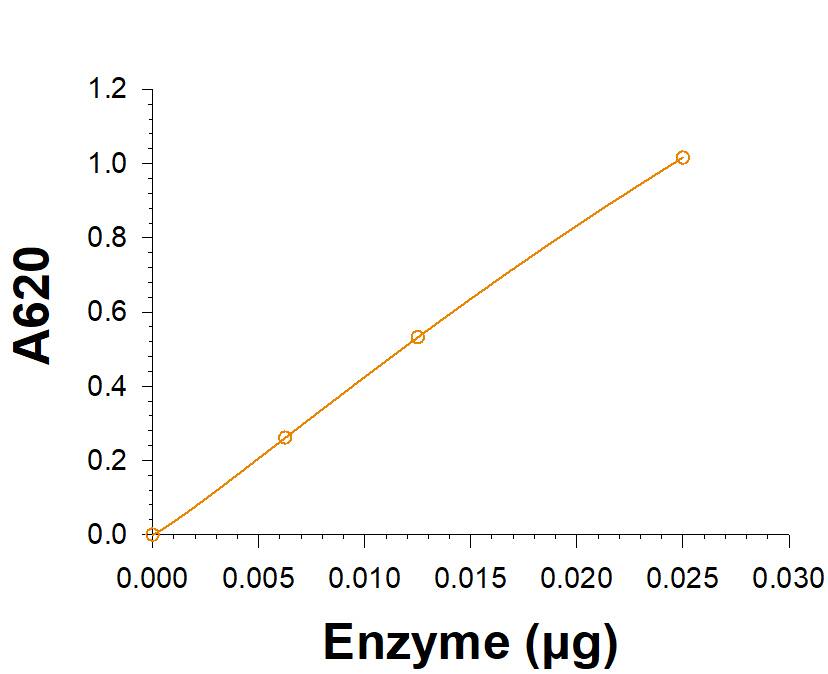
Measured by its ability to hydrolyze alpha-D-Glucose 1-phosphate.
The specific activity is >3000 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Human Muscle Glycogen Phosphorylase His-tag, CF
Recombinant Human Muscle Glycogen Phosphorylase His-tag Enzyme Activity.
Recombinant Human Muscle Glycogen Phosphorylase His-tag (Catalog # 11786-PM) is measured by its ability to hydrolyze alpha-D-Glucose 1-phosphate.Recombinant Human Muscle Glycogen Phosphorylase His-tag SDS-PAGE.
2 μg/lane of Recombinant Human Muscle Glycogen Phosphorylase His-tag (Catalog # 11786-PM) was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing bands at 86-96 kDa, under reducing conditions.Formulation, Preparation and Storage
11786-PM
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, NaCl, TCEP and Glycerol. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Glycogen phosphorylase, muscle form
Recombinant Human Muscle Glycogen Phosphorylase (PYGM), also known as myophosphorylase, is a cytoplasmic muscle isoform of glycogen phosphorylase (GP) that catalyzes the cleavage of alpha‑1,4‑glycosidic bonds in glycogen to release glucose‑1‑phosphate. PYGM is one of three mammalian isoforms known as liver, muscle, or brain GP that differ in sequence, activation regulation, kinetics, and physiological roles (1-4). PYGM is biologically active as a homodimer where each 842-residue monomer comprises N‑ and C‑terminal domains (1,3). The C-terminal domain contains a cofactor binding site for covalently bound pyridoxal cofactor. The N-terminal domain contains a key phosphorylation site that determines whether the protein is in an active or inactive state; each state is further regulated by binding of AMP, ATP, and glucose-6-phosphate in an allosteric site within the N-terminal domain. The N-terminal domain also contains a glycogen storage site. The muscle isoform PYGM is uniquely responsive to glucose for transition to an inactive state and binds AMP in a cooperative manner unique from the other isoforms. PYGM must be able to quickly respond to acute demands of energy through production of ATP for many biological processes in cells including contraction in muscles (1,3,4) and is responsive to extracellular control through neural and hormonal signals (2). PYGM is expressed in skeletal muscle but also is expressed as a predominant form of glycogen phosphorylase in the nervous system (5-7) and T lymphocytes where it binds the active form of proto-oncogenic RAC1 and leads to T-cell migration and proliferation (4,8,9). Based on regulation through extracellular signaling pathways, PYGM influences cellular processes such as signal transduction, transcription, protein stability, and cell viability through its involvement in insulin and glucagon signaling, the insulin resistance pathway, the hexosamine biosynthetic pathway where it plays a role in dynamic post-translational protein O-GlcNAcylation (7) in addition to glycogen metabolism. PYGM is also implicated and targeted as a metabolism-related oncogenic biomarker that is mis-regulated in many types of cancer including sarcoma, head and neck squamous cell carcinoma, and rectal cancer where it is often correlated with poor survival rate (4, 10-12). Finally, pathogenic dominant and recessive PYGM mutations result in glycogen accumulation and aggregates in a dominant mutation and over 200 described mutations result in autosomal recessive metabolic disease results from deficiencies of functional active enzyme known collectively as Glycogen storage disorder type V (GSDV) or McArdle disease (4,13,14). Pharmaceutical research on PYGM has focused on targeting the enzyme via gene and replacement therapy as well as small molecule treatments in McArdle disease, cardiac dysfunction, and osteosarcoma (15-18).
References
- Browner, M.F. and R.J. Fletterick. (1992) Trends. Biochem. Sci. 17:66.
- Crerar, M.M. et al. (1995) J. Biol. Chem. 270:13748.
- Lukacs, C.M. et al. (2006) Proteins. 63:1123.
- Migocka-Patrzalek, M. and M. Elias. (2021) Cells. 10:883.
- Hernández, C. et al. (2014) Acta Diabetol. 51:543.
- Jakobsen, E. et al. (2017) Neurochem. Res. 42:2490.
- Llavero, F. and J.L. Zugaza. (2024) Biochem. Soc. Trans. 52:1265.
- Arrizabalaga, O. et al. (2012) J. Biol. Chem. 287:11878.
- Llavero, F. et al. (2016) Cell Signal. 28:1713.
- Tang, Z. et al. (2017) Nucleic Acids Res. 45:W98.
- Jin, Y. and Y. Yang. (2019) Biosci. Rep. 39:BSR20191612.
- Xu, C. et al. (2025) Front. Immunol. 16:1639303.
- Llavero, F. et al. (2019) Int. J. Mol. Sci. 20:5919.
- Echaniz-Laguna, A. et al. (2020) Ann. Neurol. 88:274.
- McNamara, E.L. et al. (2020) Hum. Mol. Genet. 29:20.
- Villarreal-Salazar, M. et al. (2021) Genes (Basel). 13:74.
- Luo, L. et al. (2023) Mol. Med. 29:36.
- Gan, J. et al. (2025) Circulation. 152:1146.
Long Name
Glycogen phosphorylase, muscle form
Alternate Names
EC 2.4.1.1, Myophosphorylase, PYGM
Gene Symbol
PYGM
UniProt
Additional Glycogen phosphorylase, muscle form Products
Product Documents for Recombinant Human Muscle Glycogen Phosphorylase His-tag, CF
Product Specific Notices for Recombinant Human Muscle Glycogen Phosphorylase His-tag, CF
For research use only
Loading...
Loading...
Loading...
Loading...