Recombinant Human KGF/FGF-7 GMP Protein, CF GMP Best Seller
R&D Systems, part of Bio-Techne | Catalog # 251-GMP

Key Product Details
Product Specifications
Source
Cys32-Thr194, with an N-terminal Met
Manufactured and tested under cGMP guidelines.
Purity
Endotoxin Level
N-terminal Sequence Analysis
(Cys)32-Asn-Asp-Met-Thr-Pro-Glu-Gln-Met-Ala
Predicted Molecular Mass
SDS-PAGE
Activity
The ED50 for this effect is 6-60 ng/mL.
The specific activity of recombinant human KGF/FGF-7 is >8.16 x 105 units/mg, which is calibrated against the human KGF/FGF-7 WHO Standard (NIBSC code: 03/150).
Host Cell Protein
Mycoplasma
Host Cell DNA
Scientific Data Images for Recombinant Human KGF/FGF-7 GMP Protein, CF
Recombinant Human KGF/FGF-7 GMP Protein Bioactivity
GMP-grade Recombinant Human KGF/FGF-7 (Catalog # 251-GMP) stimulates pro-liferation of the 4MBr‑5 rhesus monkey epithelial cell line. The ED50 for this effect is 6-60 ng/mL.Recombinant Human KGF/FGF-7 GMP Protein SDS-PAGE
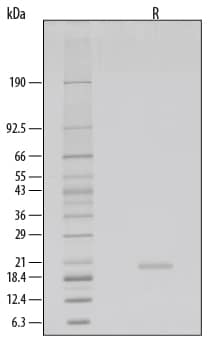
1 μg/lane of GMP-grade Recombinant Human KGF/FGF-7 (Catalog # 251-GMP) was resolved with SDS-PAGE under reducing (R) conditions and visualized by silver staining, showing a single band at 20 kDa.Formulation, Preparation and Storage
251-GMP
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 100 μg/mL in PBS. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: KGF/FGF-7
KGF (keratinocyte growth factor), also known as FGF-7 (fibroblast growth factor-7), is one of 22 known members of the mouse FGF family of secreted proteins that plays a key role in development, morphogenesis, angiogenesis, wound healing, and tumorigenesis (1-4). KGF expression is restricted to cells of mesenchymal origin. When secreted, it acts as a paracrine growth factor for nearby epithelial cells (1). KGF speeds wound healing by being dramatically upregulated in response to damage to skin or internal structures that results in high local concentrations of inflammatory mediators such as IL-1 and TNF-alpha. (2, 5). KGF promotes cell migration and invasion, and mediates melanocyte transfer to keratinocytes upon UVB radiation (6, 7). It has been used ectopically to avoid chemotherapy-induced oral mucositis in patients with hematological malignancies (1). Deletion of KGF affects kidney development, producing abnormally small ureteric buds and fewer nephrons (8). It also impedes hair follicle differentiation (9). The 194 amino acid (aa) KGF precursor contains a 31 aa signal sequence and, like all other FGFs, an ~120 aa beta-trefoil scaffold that includes receptor- and heparin-binding sites. KGF signals only through the IIIb splice form of the tyrosine kinase receptor, FGF R2 (FGF R2-IIIb/KGF R) (10). Receptor dimerization requires an octameric or larger heparin or heparin sulfate proteoglycan (11). FGF-10, also called KGF2, shares 51% aa identity and similar function to KGF, but shows more limited expression than KGF and uses an additional receptor, FGF R2-IIIc (12). Following receptor engagement, KGF is typically degraded, while FGF-10 is recycled (12). Mature human KGF, which is active across species, shares 98% aa sequence identity with bovine, equine, ovine and canine, 96% with mouse and porcine, and 92% with rat KGF, respectively.
References
- Finch, P.W. and J.S. Rubin (2006) J. Natl. Cancer Inst. 98:812.
- Werner, S. et al. (2007) J. Invest. Dermatol. 127:998.
- Werner, S. (1998) Cytokine Growth Factor Rev. 9:153.
- Mason, I.J. et al. (1994) Mech. Dev. 45:15.
- Geer, D.J. et al. (2005) Am. J. Pathol. 167:1575.
- Niu, J. et al. (2007) J. Biol. Chem. 282:6001.
- Cardinali, G. et al. (2005) J. Invest. Dermatol. 125:1190.
- Qiao, J. et al. (1999) Development 126:547.
- Guo, L. et al. (1996) Genes Dev. 10:165.
- de Georgi, V. et al. (2007) Dermatol. Clin. 25:477.
- Hsu, Y-R. et al. (1999) Biochemistry 38:2523.
- Belleudi, F. et al. (2007) Traffic 8:1854.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional KGF/FGF-7 Products
Product Documents for Recombinant Human KGF/FGF-7 GMP Protein, CF
Manufacturing Specifications
GMP ProteinsR&D Systems, a Bio-Techne Brand's GMP proteins are produced according to relevant sections of the following documents: USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Eu. Ph. 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products.
R&D Systems' quality focus includes:
- Designed, manufactured and tested under an ISO 9001:2015 and ISO 13485:2016 certified quality system
- Documented and controlled manufacturing process
- Control of documentation and process changes by QA
- Personnel training programs
- Raw material inspection and vendor qualification/monitoring program
- Validated equipment, processes and test methods
- Equipment calibration and maintenance schedules using a Regulatory Asset Manager
- Facility/Utilities maintenance, contamination controls, safety and pest control programs
- Material review process for variances
- Robust product stability program following relevant ICH guidelines
- N-terminal amino acid analysis
- SDS-PAGE purity analysis
- Molecular weight analysis via mass spectrometry
- Endotoxin assessment per USP <85> and Ph. Eur. 2.6.14 guidelines
- Bioassay analysis
- Microbial testing per USP <71> and Ph. Eur. 2.6.1 guidelines
- Host cell protein assessment
- Host cell DNA assessment
- Mycoplasma assessment
Production records and facilities are available for examination by appropriate personnel on-site at R&D Systems in Minneapolis and St. Paul, Minnesota USA.
R&D Systems sells GMP grade products for preclinical or clinical ex vivo use. They are not for in vivo use. Please read the following End User Terms prior to using this product.
Product Specific Notices for Recombinant Human KGF/FGF-7 GMP Protein, CF
Full terms and conditions of sale can be found online in the Protein Sciences Segment T&Cs at: Terms & Conditions.
For preclinical, or clinical ex vivo use