Recombinant Human FGF basic/FGF2 (146 aa) GMP Protein, CF GMP
R&D Systems, part of Bio-Techne | Catalog # 233-GMP

Key Product Details
Product Specifications
Source
Pro143-Ser288, with an N-terminal Ala
Produced using non-animal reagents in an animal-free laboratory.
Manufactured and tested under cGMP guidelines.
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The ED50 for this effect is 0.1-0.6 ng/mL.
The specific activity of Recombinant Human FGF basic/FGF2 GMP is >8.0 x 105 IU/mg, which is calibrated against the human FGF basic/FGF2 WHO International Standard (NIBSC code: 90/712).
Host Cell Protein
Mycoplasma
Host Cell DNA
Reviewed Applications
Read 1 review rated 5 using 233-GMP in the following applications:
Scientific Data Images for Recombinant Human FGF basic/FGF2 (146 aa) GMP Protein, CF
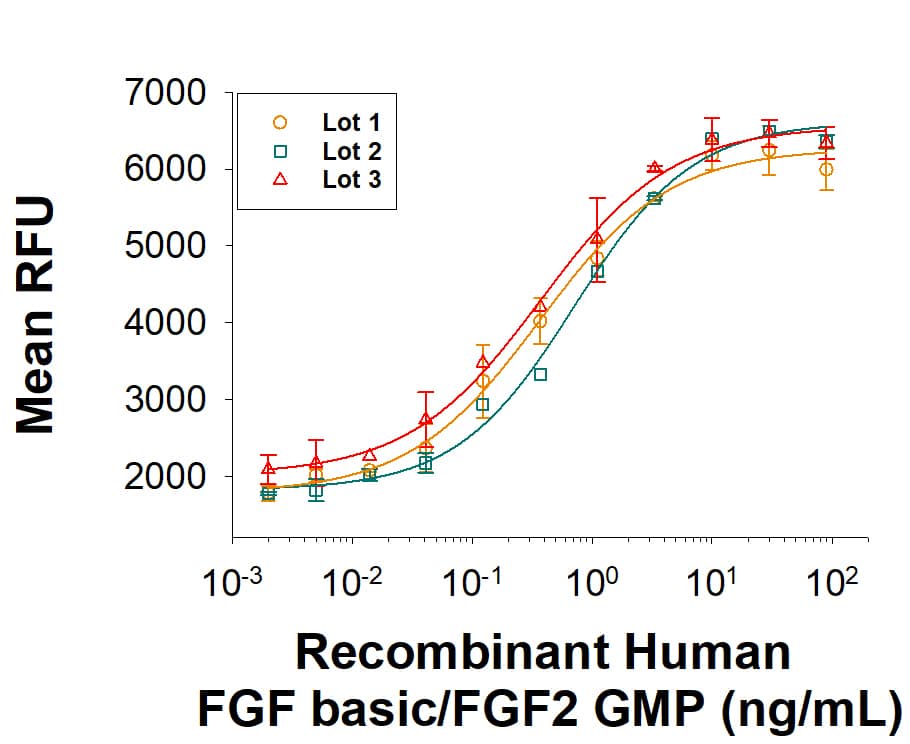
Recombinant Human FGF basic/FGF2 (146 aa) GMP Protein Bioactivity
GMP‑grade Recombinant Human FGF basic/FGF2 (Catalog # 233-GMP) stimulates proliferation of the NR6R‑3T3 mouse fibroblast cell line. The ED50 is 0.1-0.6 ng/mL. Three independent lots were tested for activity and plotted on the same graph to show lot-to-lot consistency of GMP FGF basic/FGF2.Recombinant Human FGF basic/FGF2 (146 aa) GMP Protein SDS-PAGE
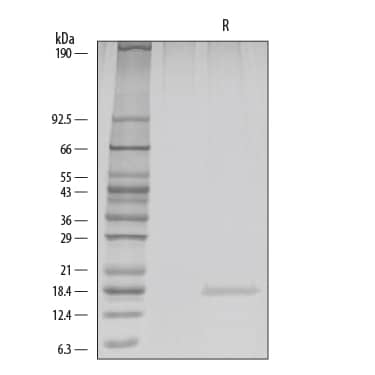
1 µg/lane of GMP-grade Recombinant Human FGF basic/FGF2 (Catalog # 233-GMP) was resolved by SDS-PAGE with silver staining, under reducing (R) conditions, showing a single band at 17 kDa.Equivalent Bioactivity of GMP and Animal-Free grades of Recombinant Human FGF basic
Equivalent bioactivity of GMP (Catalog # 233-GMP) and Animal-Free (AFL233) grades of Recombinant Human FGF basic as measured in a cell proliferation assay using NR6R‑3T3 mouse fibroblast cells (orange and green, respectively).Formulation, Preparation and Storage
233-GMP
| Formulation | Lyophilized from a 0.2 μm filtered solution in Tris-HCl and NaCl. |
| Reconstitution | Reconstitute at 100 μg/mL in PBS. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: FGF basic/FGF2/bFGF
FGF basic (also known as FGF2 and HBGF-2) is a member of the FGF family of at least 23 related mitogenic proteins which show 35-60% amino acid conservation. FGF acidic and basic, unlike the other members of the family, lack signal peptides and are apparently secreted by mechanisms other than the classical protein secretion pathway. FGF basic has been isolated from a number of sources, including neural tissue, pituitary, adrenal cortex, corpus luteum, and placenta. This factor contains four cysteine residues, but reduced FGF basic retains full biological activity, indicating that disulfide bonds are not required for this activity. A variety of forms of FGF basic are produced as a result of N-terminal extensions. These extensions affect localization of FGF basic in cellular compartments but do not affect biological activity. Binding of FGF to heparin or cell surface heparan sulfate proteoglycans is necessary for binding of FGF to high affinity FGF receptors. FGF acidic and basic appear to bind to the same high affinity receptors and show a similar range of biological activities. FGF basic stimulates the proliferation of all cells of mesodermal origin and many cells of neuroectodermal, ectodermal, and endodermal origin. FGF basic induces neuron differentiation, survival, and regeneration. FGF basic also modulates embryonic development and differentiation. These observed in vitro functions of FGF basic suggest FGF basic may play a role in vivo in the modulation of such normal processes as angiogenesis, wound healing and tissue repair, embryonic development and differentiation, and neuronal function and neural degeneration. Additionally, FGF basic may participate in the production of a variety of pathological conditions resulting from excessive cell proliferation and excessive angiogenesis.
References
- Coulier, F. et al. (1997) J. Mol. Evol. 44:43.
- Chen, C.H. et al. (2004) Curr. Vasc. Pharmacol. 2:33.
- Mohammadi, M. et al. (2005) Curr. Opin. Struct. Biol. 15:506.
- Fernig, D. et al. (1994) Prog. Growth Factor Res. 5:353.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional FGF basic/FGF2/bFGF Products
Product Documents for Recombinant Human FGF basic/FGF2 (146 aa) GMP Protein, CF
Manufacturing Specifications
GMP ProteinsR&D Systems, a Bio-Techne Brand's GMP proteins are produced according to relevant sections of the following documents: USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Eu. Ph. 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products.
R&D Systems' quality focus includes:
- Manufactured and tested under an ISO 9001:2015 and ISO 13485:2016 certified quality system
- Documented processes and QA control of documentation and process changes
- Personnel training programs
- Raw material testing and vendor qualification/monitoring
- Fully validated equipment, processes and test methods
- Equipment calibration schedules using a computerized calibration program
- Facility maintenance, safety programs and pest control
- Material review process for variances
- Monitoring of stability over product shelf-life
- N-terminal amino acid analysis, SDS-PAGE analysis, mass spectrometry results, and endotoxin level (as determined by LAL assay) performed on each bulk QC lot, not on individual bottlings of each QC lot
- Post-bottling lot-specific bioassay results (compliance with an established range) and results of microbial testing according to USP
- Host Cell Protein testing performed by ELISA
- Mycoplasma testing by ribosomal RNA hybridization assay
Production records and facilities are available for examination by appropriate personnel on-site at R&D Systems in Minneapolis, Minnesota USA.
R&D Systems sells GMP grade products for preclinical or clinical ex vivo use. They are not for in vivo use. Please read the following End User Terms prior to using this product.
Animal-Free Manufacturing Conditions
Our dedicated controlled-access animal-free laboratories ensure that at no point in production are the products exposed to potential contamination by animal components or byproducts. Every stage of manufacturing is conducted in compliance with R&D Systems' stringent Standard Operating Procedures (SOPs). Production and purification procedures use equipment and media that are confirmed animal-free.
Production
- All molecular biology procedures use animal-free media and dedicated labware.
- Dedicated fermentors are utilized in committed animal-free areas.
- Protein purification columns are animal-free.
- Bulk proteins are filtered using animal-free filters.
- Purified proteins are stored in animal-free containers in a dedicated cold storage room.
Quality Assurance
- Low Endotoxin Level.
- No impairment of biological activity.
- High quality product obtained under stringent conditions.
Product Specific Notices for Recombinant Human FGF basic/FGF2 (146 aa) GMP Protein, CF
Full terms and conditions of sale can be found online in the Protein Sciences Segment T&Cs at: Terms & Conditions.
For preclinical, or clinical ex vivo use