Lysozyme Antibody - BSA Free
Novus Biologicals, part of Bio-Techne | Catalog # NBP2-61118

![Immunocytochemistry/ Immunofluorescence: Lysozyme Antibody - BSA Free [NBP2-61118] Immunocytochemistry/ Immunofluorescence: Lysozyme Antibody - BSA Free [NBP2-61118]](https://resources.bio-techne.com/images/products/Lysozyme-Antibody-Immunocytochemistry-Immunofluorescence-NBP2-61118-img0006.jpg)
Conjugate
Catalog #
Key Product Details
Species Reactivity
Validated:
Human, Mouse
Cited:
Mouse
Applications
Validated:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Flow Cytometry, Immunocytochemistry/ Immunofluorescence, Simple Western
Cited:
Immunohistochemistry-Paraffin, Western Blot, Immunocytochemistry/ Immunofluorescence
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Format
BSA Free
Concentration
1.0 mg/ml
Product Specifications
Immunogen
Full length recombinant human Lysozyme protein.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for Lysozyme Antibody - BSA Free
Immunocytochemistry/ Immunofluorescence: Lysozyme Antibody - BSA Free [NBP2-61118]
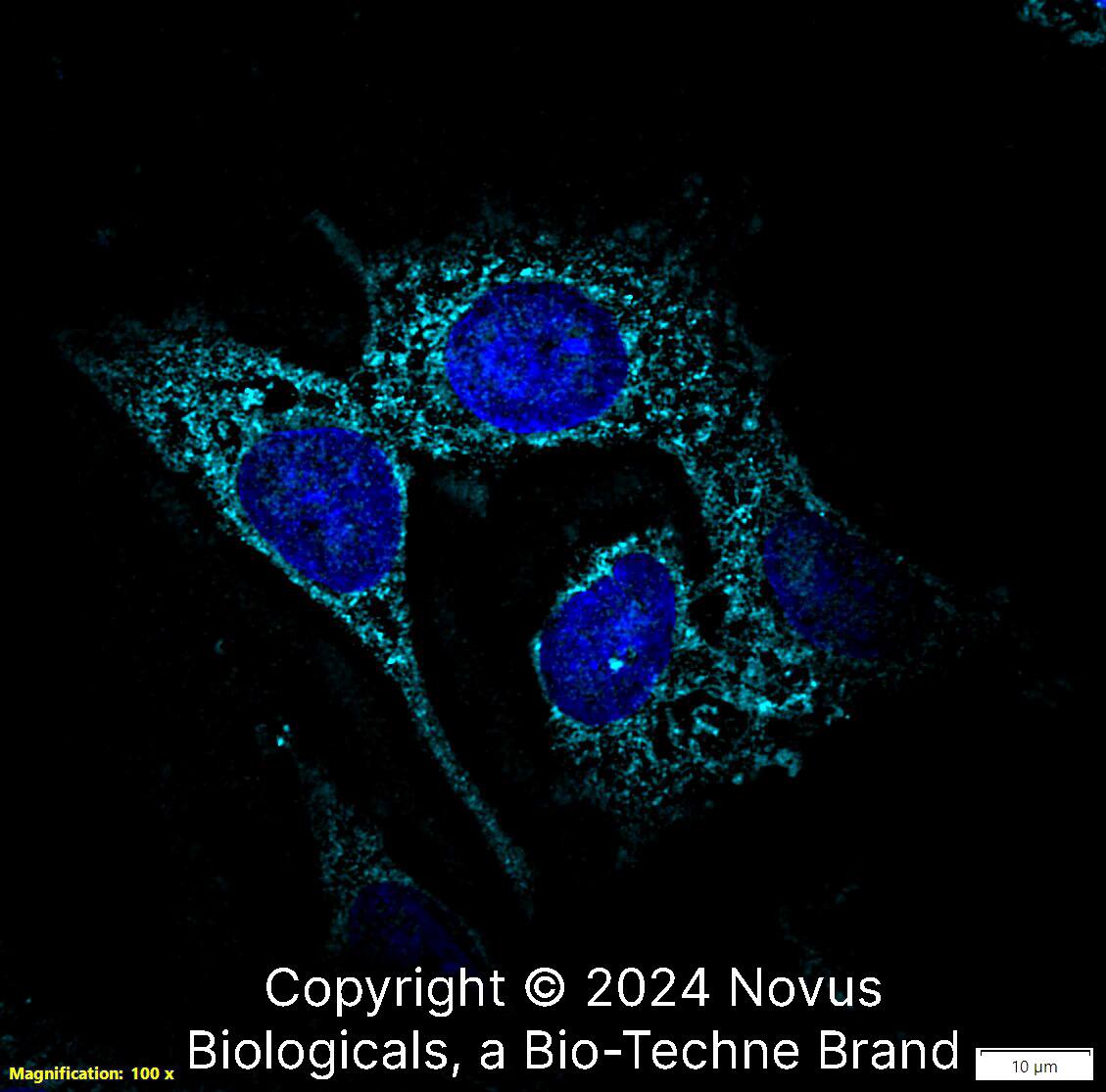
Immunocytochemistry/Immunofluorescence: Lysozyme Antibody [NBP2-61118] - NIH3T3 cells were fixed and permeabilized for 10 minutes using -20C MeOH. The cells were incubated with anti-Lysozyme NBP2-61118 at 2 ug/ml overnight at 4C and detected with an anti-rabbit Dylight 488 (Green) at a 1:1000 dilution for 60 minutes. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 100X objective and digitally deconvolved.Western Blot: Lysozyme AntibodyBSA Free [NBP2-61118]
Western Blot: Lysozyme Antibody [NBP2-61118] - Total protein from human cell lines THP-1 and HepG2, human stomach and kidney as well as mouse kidney was separated on a 4-20% gel by SDS-PAGE, transferred to 0.2 um PVDF membrane and blocked in 5% non-fat milk in TBST. The membrane was probed with 2.0 ug/ml anti-Lysozyme in 5% non-fat milk in TBST and detected with an anti-rabbit HRP secondary antibody using chemiluminescence.Immunohistochemistry-Paraffin: Lysozyme Antibody - BSA Free [NBP2-61118]
Immunohistochemistry-Paraffin: Lysozyme Antibody [NBP2-61118] - Analysis of a FFPE tissue section of human kidney with Lysozyme antibody at 1:100 dilution. The staining was developed with HRP-DAB detection method and the counterstaining was performed using hematoxylin. This Lysozyme antibody generated an expected cytoplasmic staining in all the cells with strongest signal in tubular epithelial cells. Some tubules showed more of a punctate staining pattern (vesicular Lysozyme) while all of the tubules depicted a diffused signal (secreted Lysozyme). A subset of cells, especially those from glomeruli, showed nuclear positivity also.Applications for Lysozyme Antibody - BSA Free
Application
Recommended Usage
Immunocytochemistry/ Immunofluorescence
2 ug/ml
Immunohistochemistry
1:100 - 1:250
Immunohistochemistry-Paraffin
1:100 - 1:250
Western Blot
2 ug/ml
Formulation, Preparation, and Storage
Purification
Immunogen affinity purified
Formulation
PBS
Format
BSA Free
Preservative
0.02% Sodium Azide
Concentration
1.0 mg/ml
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles.
Background: Lysozyme
Alternate Names
EC 3.2.1.17, lysozyme, lysozyme (renal amyloidosis)1,4-beta-N-acetylmuramidase C, lysozyme C, LZM, renal amyloidosis
Gene Symbol
LYZ
Additional Lysozyme Products
Product Documents for Lysozyme Antibody - BSA Free
Product Specific Notices for Lysozyme Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Loading...
Loading...
Loading...
Loading...
Loading...
![Western Blot: Lysozyme AntibodyBSA Free [NBP2-61118] Western Blot: Lysozyme AntibodyBSA Free [NBP2-61118]](https://resources.bio-techne.com/images/products/Lysozyme-Antibody-Western-Blot-NBP2-61118-img0003.jpg)
![Immunohistochemistry-Paraffin: Lysozyme Antibody - BSA Free [NBP2-61118] Immunohistochemistry-Paraffin: Lysozyme Antibody - BSA Free [NBP2-61118]](https://resources.bio-techne.com/images/products/Lysozyme-Antibody-Immunohistochemistry-Paraffin-NBP2-61118-img0001.jpg)
![Flow Cytometry: Lysozyme Antibody - BSA Free [NBP2-61118] Flow Cytometry: Lysozyme Antibody - BSA Free [NBP2-61118]](https://resources.bio-techne.com/images/products/Lysozyme-Antibody-Flow-Cytometry-NBP2-61118-img0007.jpg)
![Immunocytochemistry/ Immunofluorescence: Lysozyme Antibody - BSA Free [NBP2-61118] Immunocytochemistry/ Immunofluorescence: Lysozyme Antibody - BSA Free [NBP2-61118]](https://resources.bio-techne.com/images/products/Lysozyme-Antibody-Immunocytochemistry-Immunofluorescence-NBP2-61118-img0002.jpg)
![Flow Cytometry: Lysozyme Antibody - BSA Free [NBP2-61118] Flow Cytometry: Lysozyme Antibody - BSA Free [NBP2-61118]](https://resources.bio-techne.com/images/products/Lysozyme-Antibody-Flow-Cytometry-NBP2-61118-img0005.jpg)
![Simple Western: Lysozyme AntibodyBSA Free [NBP2-61118] Simple Western: Lysozyme AntibodyBSA Free [NBP2-61118]](https://resources.bio-techne.com/images/products/Lysozyme-Antibody-Simple-Western-NBP2-61118-img0004.jpg)