Human Fas/TNFRSF6/CD95 Antibody
R&D Systems, part of Bio-Techne | Catalog # MAB142

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Fas/TNFRSF6/CD95 Antibody
Detection of Fas/TNFRSF6/CD95 by Flow Cytometry
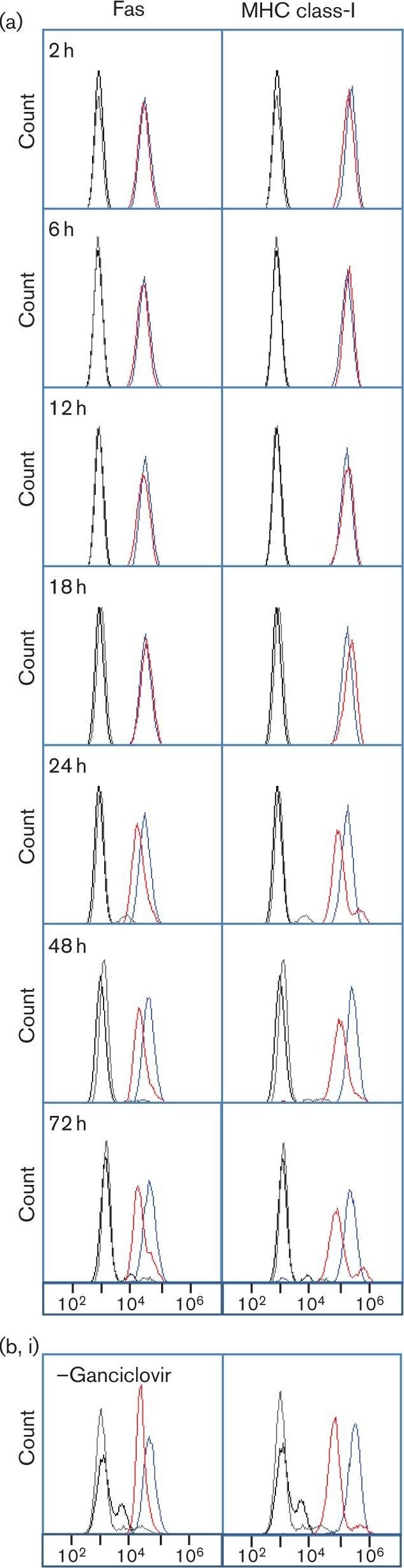
Modulation of Fas cell surface expression in cells infected with HCMV. (a) HFFF-hTERTs were infected with HCMV strain Merlin (m.o.i. 10) or mock-infected, and analysed at indicated time points by flow cytometry for cell surface Fas expression [mAb142 (R&D Systems), n≥3). (b) HFFFs were infected with HCMV strain Merlin (m.o.i. 10, 72 h) in the presence (i) or absence (ii) of 100 µM ganciclovir and analysed by flow cytometry for cell surface Fas expression (n = 3). (c) HFFF-hTERTs were incubated with supernatants (sn) of strain Merlin-infected cells (m.o.i. 10, 72 h p.i.) from which virions had been removed using a 0.1 µm filter, or were infected with HCMV strain Merlin (m.o.i. 10, 72 h) or an equivalent gamma-irradiated preparation (2500 Gy) and analysed by flow cytometry for cell surface Fas expression (n = 3). (d) HFFF-hTERTs were infected with HCMV strains Merlin, AD169, FIX or TB40 (m.o.i. 10, 72 h) and analysed by flow cytometry for cell surface Fas expression (n≥3). Control IgG staining is denoted by black and grey lines for mock-infected and HCMV-infected cells, respectively. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/24394698), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Fas/TNFRSF6/CD95 Antibody
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human Alzheimer's disease brain
Western Blot
Sample: Recombinant Human Fas/TNFRSF6/CD95 Fc Chimera (Catalog # 326-FS)
under non-reducing conditions only
Reviewed Applications
Read 1 review rated 5 using MAB142 in the following applications:
Formulation, Preparation, and Storage
Purification
Reconstitution
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Background: Fas/TNFRSF6/CD95
Fas, also known as APO-1, CD95, and TNFRSF6, was originally identified as a cell-surface protein which binds to monoclonal antibodies that were cytolytic for various human cell lines. In the TNF Receptor superfamily nomenclature, Fas is referred to as TNFRSF6. Human Fas cDNA encodes a 325 amino acid (aa) residue type 1 membrane protein that belongs to the TNF and NGF receptor family. Alternatively spliced cDNAs encoding multiple Fas isoforms, including a soluble form of Fas lacking the transmembrane domain, have also been identified. Fas is highly expressed in epithelial cells, hepatocytes, activated mature lymphocytes, virus-transformed lymphocytes, and other tumor cells. Fas expression has also been detected in mouse thymus, liver, heart, lung, kidney, and ovary. The ligand for Fas (FasL) has been identified and shown to be a member of the TNF family of type 2 membrane proteins. FasL is predominantly expressed by activated T‑lymphocytes, NK cells, and in tissues with immune-privileged sites. Soluble FasL can be produced by proteolysis of membrane-associated Fas.
Ligation of Fas by FasL or anti-Fas antibody has been shown to induce apoptotic cell death in Fas-bearing cells. Fas plays a role in the down-regulation of the immune reaction and has been shown to be a key mediator of activation-induced death of activated T lymphocytes. Fas-mediated cell death has also been shown to be important for the deletion of activated or autoreactive B lymphocytes. Besides the perforin/granzyme-based mechanism, the Fas system has been identified as the alternate pathway for CTL-mediated cytotoxicity. FasL has also been shown to function in immunological privileged sites by killing infiltrating Fas-bearing lymphocytes and inflammatory cells.
References
- Nagata, S. and P. Golstein (1995) Science 267:1449.
- Nagata, S. (1997) Cell 88:355.
- Parijs, L. and A.K. Abbas (1996) Current Opinion in Immunol. 8:355.
- Green, D.R. and C.F. Ware (1997) Proc. Natl. Acad. Sci. USA 94:5986.
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
Additional Fas/TNFRSF6/CD95 Products
Product Documents for Human Fas/TNFRSF6/CD95 Antibody
Product Specific Notices for Human Fas/TNFRSF6/CD95 Antibody
For research use only