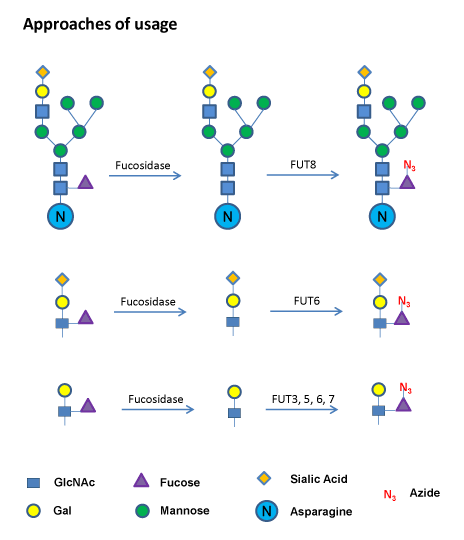
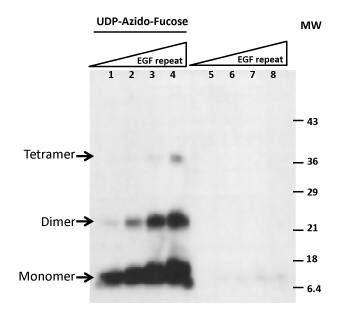
GDP-Azido-Fucose
R&D Systems, part of Bio-Techne | Catalog # ES101
Activated Sugar

Key Product Details
| Key Benefits: |
Learn more about Fluorescent Glycan Labeling and Detection |
Assay Procedure
Sample Protocol for Core-6 Fucose Labeling
Protocols are guidelines. Parameters need to be optimized by end users.
Materials
- Assay Buffer: 25 mM HEPES, 150 mM NaCl, 10 mM MnCl2, pH 7.5.
- Protein Sample
- Recombinant Human Fucosyltransferase 8/FUT8 (R&D Systems, Catalog # 5768-GT)
- GDP-Azido-Fucose (R&D Systems, Catalog # ES101)
- Biotinylated Alkyne (R&D Systems, Catalog # ES100)
- CuCl2, 1 mM in deionized water
- Ascorbic Acid, 20 mM in deionized water
- SDS-PAGE and Western blot reagents or equivalent
- TBST buffer: 25 mM Tris, 137 mM NaCl, 0.1% Tween-20, pH 7.5
- Streptavidin-HRP (R&D Systems, Catalog # DY998)
Assay Procedure
- Prepare a reaction mixture by combining 5 µg of Protein Sample, 1 µg of rhFUT8 in the presence of 1 nmol GDP-Azido-Fucose in the Assay Buffer with the final volume of 25 µL.
- Prepare negative controls according to step 1 but omit Protein Sample or rhFUT8 or GDP-Azido-Fucose.
- Incubate all the reactions and the controls at 37°C for one hour.
- Add to each of the samples: 5 µL of 1 mM CuCl2, 5 µL of 20 mM Ascorbic Acid, and 5 µL of 1 mM Biotinylated Alkyne. Mix with gentle tapping.
- Incubate all samples at room temperature for 1 hour.
- Separate the reactions and controls by 12% SDS-PAGE.
- Blot the gel to a nitrocellulose membrane.
- Block the blot with 10% fat-free milk for 5 minutes.
- Thoroughly wash the membrane with TBST buffer by changing buffer three times for a total of 45 minutes.
- Incubate the blot with 25 ng/mL Streptavidin-HRP in 30 mL TBST buffer for 30 minutes.
- Thoroughly wash the membrane with TBST buffer by changing buffer three times for a total of 45 minutes.
- Detect with commercial ECL (Enhanced Chemiluminescence) reagents.
Final Assay Conditions Per Reaction
- GDP-Azido-Fucose: 1 nmol
- rhFUT8: 1 µg
- Protein Sample: 5 µg
- Reaction volume: 25 µl
Click Chemistry Reaction Conditions Per Reaction
- CuCl2: 5 nmol
- Ascorbic Acid: 100 nmol
- Biotinylated Alkyne: 5 nmol
- Reaction volume: 40 µl
Loading...
Customer Reviews for GDP-Azido-Fucose
There are currently no reviews for this product. Be the first to review GDP-Azido-Fucose and earn rewards!
Have you used GDP-Azido-Fucose?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review