Recombinant Human GGT1 His-tag Protein, CF
R&D Systems, part of Bio-Techne | Catalog # 10977-GT

Key Product Details
- R&D Systems CHO-derived Recombinant Human GGT1 His-tag Protein (10977-GT)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Product Specifications
Source
Chinese Hamster Ovary cell line, CHO-derived human gamma-Glutamyltransferase 1/GGT1 protein
Pro27-Tyr569 with a C-terminal 6-His
Pro27-Tyr569 with a C-terminal 6-His
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Pro27 & Thr381
Predicted Molecular Mass
39 kDa (heavy chain) & 21 kDa (light chain)
SDS-PAGE
56-69 kDa & 22-26 kDa, under reducing conditions
Activity
Measured by its ability to hydrolyze glutathione to Glu and Cys-Gly.
The specific activity is >1300 pmol/min/μg, as measured under the described conditions.
The specific activity is >1300 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Human GGT1 His-tag Protein, CF
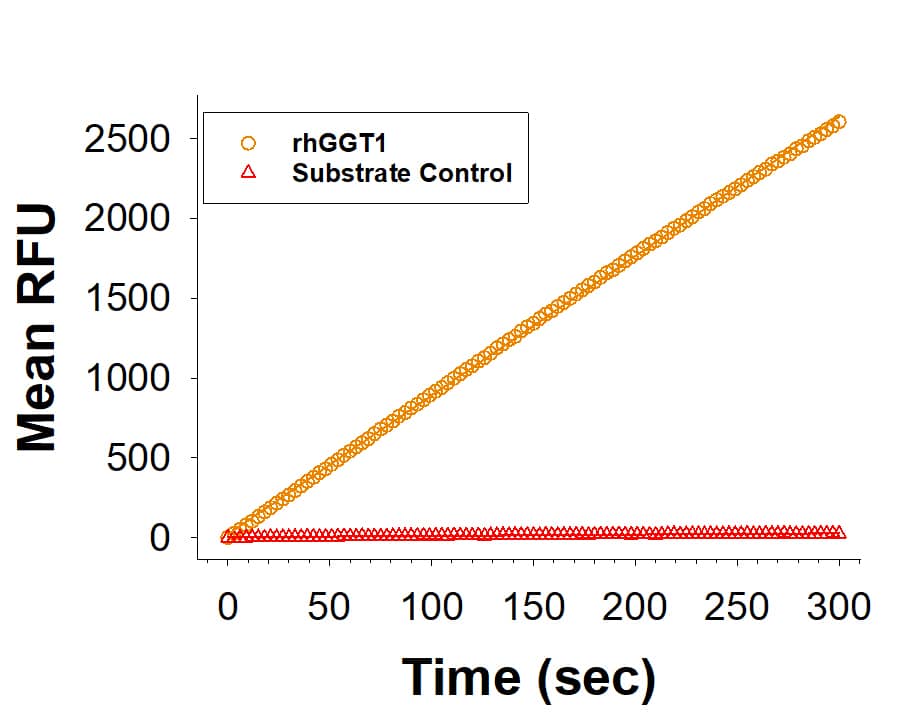
Recombinant Human GGT1 His-tag Protein Enzyme Activiy.
Recombinant Human GGT1 His-tag (Catalog # 10977-GT) is measured by its ability to hydrolyze glutathione to Glu and Cys-Gly.Recombinant Human GGT1 His-tag Protein SDS-PAGE.
2 μg/lane of Recombinant Human GGT1 His-tag Protein (Catalog # 10977-GT) was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing bands at 56-69 kDa and 22-26 kDa, under reducing conditions.Formulation, Preparation and Storage
10977-GT
| Formulation | Supplied as a 0.2 μm filtered solution in Tris and NaCl. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: gamma-Glutamyltransferase 1/GGT1
References
- Pawlak, A. et al. (1988) J. Biol. Chem. 263:9913.
- Wickham, S. et al. (2011) Anal. Biochem. 414:208.
- Heisterkamp, N. et al. (1991) Proc. Natl. Acad. Sci. USA 88:6303.
- West, M.B. et al. (2013) J Biol. Chem. 288:31902.
- Ndrepepa, G. et al. (2018) Clin. Chim. Acta. 476:130.
- Lieberman, M.W. et al. (1996) Proc. Natl. Acad. Sci. 93: 7923.
- Hanigan, M.H. (2014) Adv. Cancer Res. 122:103.
- Corti, A. et al. (2020) Free Radic. Biol. Med.160:807.
- Sian, J. et al. (1994) Ann. Neurol. 36:356.
- Whitfield, J. B. (2001) Crit. Rev. Clin. Lab Sci. 38:263.
- Lyu, S.C. et al. (2021) Cancer Manag. Res. 13:4887.
- Carr, B.I. et al. (2021) J. Transl. Sci. 7:10.15761.
- Sayan, M. et al. (2021) Wiad. Lek. 74: 1804.
- Jia, Y. et al. (2021) J. Oncol. 9120265.
Long Name
gamma-Glutamyltransferase 1
Alternate Names
CD224, D22S672, D22S73, GGT, GGT-1, GGTD
Entrez Gene IDs
2678 (Human)
Gene Symbol
GGT1
UniProt
Additional gamma-Glutamyltransferase 1/GGT1 Products
Product Documents for Recombinant Human GGT1 His-tag Protein, CF
Product Specific Notices for Recombinant Human GGT1 His-tag Protein, CF
For research use only
Loading...
Loading...
Loading...