Recombinant Human F13A1 His-tag Protein, CF
R&D Systems, part of Bio-Techne | Catalog # 10179-F1

Key Product Details
- R&D Systems Sf 21 (baculovirus)-derived Recombinant Human F13A1 His-tag Protein (10179-F1)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Sf 21 (baculovirus)
Accession #
Conjugate
Unconjugated
Applications
Enzyme Activity
Product Specifications
Source
Spodoptera frugiperda, Sf 21 (baculovirus)-derived human F13A1 protein
Ser2-Met732
with an N-terminal Met and 6-His tag
Ser2-Met732
with an N-terminal Met and 6-His tag
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
No results obtained, Met predicted. Protein identity confirmed by MS analysis of tryptic fragments.
Predicted Molecular Mass
84 kDa
SDS-PAGE
76-88 kDa, under reducing conditions
Activity
Measured by its ability to release DNP from Abz-NE(CAD-DNP)EQVSPLTLLK-OH.
The specific activity is >13.0 pmol/min/μg, as measured under the described conditions.
The specific activity is >13.0 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Human F13A1 His-tag Protein, CF
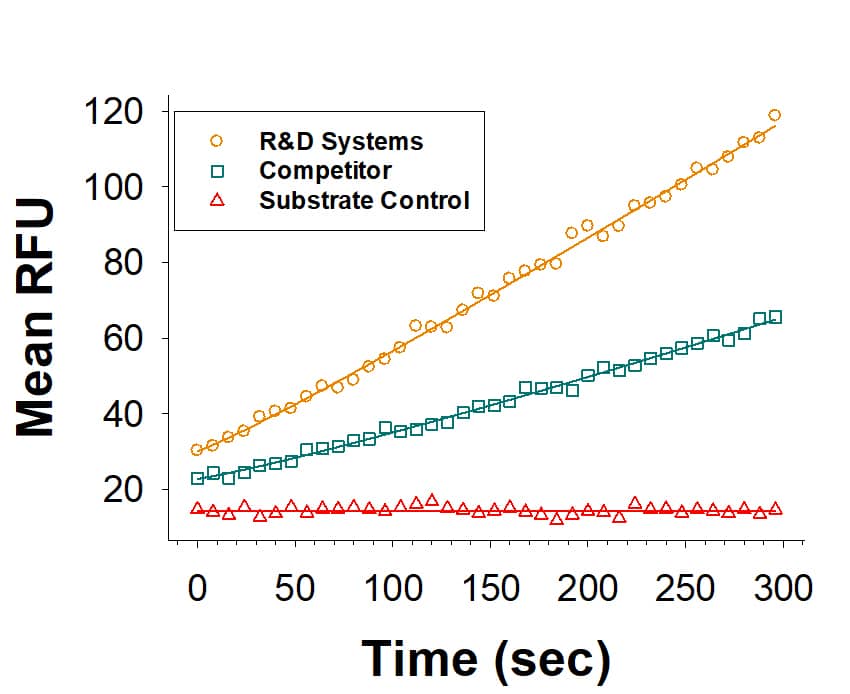
Recombinant Human F13A1 His-tag Protein Enzyme Activity
Recombinant Human F13A1 His-tag (Catalog # 10179-F1) is measured by its ability to release DNP from Abz-NE(CAD-DNP)EQVSPLTLLK-OH. The activity (orange) is approximately 2-fold higher than the competitor's F13A1 (green).Recombinant Human F13A1 His-tag Protein SDS-PAGE
2 μg/lane of Recombinant Human F13A1 His-tag was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® blue staining, showing a band at ~82 kDa under reducing conditions.Formulation, Preparation and Storage
10179-F1
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, NaCl, EDTA, Glycerol and TCEP. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: F13A1
References
- Griffin, M. et al. (2002) Biochem. J. 368:377.
- Muszbek, L. et al. (1999) Thromb. Res. 94:271.
- Yee, V. C. et al. (1994) Proc. Natl. Acad. Sci. USA 91:7296.
- Lord, S. T. et al. (2011) Aterioscler. Thromb. Vasc. Biol. 31:494.
- Fraser, S. R. et al. (2011) Blood 117:6371.
- Nikolajsen, C. L. et al. (2014) J. Biol. Chem. 289:6526.
- AbdAlla, S. et al. (2004) Cell. 119:343.
- Nahrendorf, M. et al. (2006) Circulation 113:1196.
- Dardik, R. et al. (2006) Thromb. Haemost. 95:546.
- Dardik, R. et al. (2005) Arterioscler. Thromb. Vasc. Biol. 25:526
- Asahina, T. et al. (2000) Placenta. 21:388.
- Piercy-Kotb, S. A. et al. (2012) J. Cell Physiol. 227:2936.
- Richardson, V. R. et al. (2012) Br. J. Haematol. 160:116.
- Simon, A. et al. (2012) Cytometry B. Clin. Cytom. 82:209.
- Naderi, M. et al. (2015) Hematology. 20:112.
- Inbal, A. et al. (2005) Thromb. Haemost. 94:432.
- Inbal, A. and L. Muszbek. (2003) Semin. Thromb. Hemost. 29:171
- Naderi, M. et al. (2016) Iran J. Pharm. Res. 15:635.
Long Name
Coagulation Factor XIII, A1 Polypeptide
Alternate Names
F13A, FSF, A Subunit
Entrez Gene IDs
2162 (Human)
Gene Symbol
F13A1
UniProt
Additional F13A1 Products
Product Documents for Recombinant Human F13A1 His-tag Protein, CF
Product Specific Notices for Recombinant Human F13A1 His-tag Protein, CF
For research use only
Loading...
Loading...
Loading...