Recombinant Human CD302/CLEC13A Fc Chimera Protein, CF
R&D Systems, part of Bio-Techne | Catalog # 10203-CL

Key Product Details
- R&D Systems HEK293-derived Recombinant Human CD302/CLEC13A Fc Chimera Protein (10203-CL)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
HEK293
Accession #
Structure / Form
Disulfide-linked homodimer
Conjugate
Unconjugated
Applications
Bioactivity
Product Specifications
Source
Human embryonic kidney cell, HEK293-derived human CD302/CLEC13A protein
| Human CD302/CLEC13A (Asp23-Asn167) Accession # Q8IX05-1 |
IEGRMD | Human IgG1 (Pro100-Lys330) |
| N-terminus | C-terminus |
Purity
>90%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Asp23
Predicted Molecular Mass
43 kDa
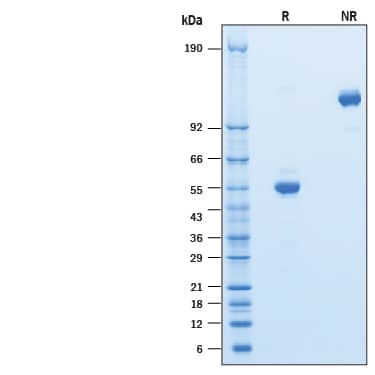
SDS-PAGE
51-59 kDa, under reducing conditions
Activity
Measured by its binding ability in a functional ELISA.
When Recombinant Human CD302/CLEC13A Fc Chimer (Catalog # 10203-CL) is immobilized at 5 μg/mL, 100 μL/well, the concentration of Recombinant Human DEC-205/CD205 Fc Chimera (Catalog # 10205-DE) that produces 50% of the optimal binding response is 4-24 μg/mL.
When Recombinant Human CD302/CLEC13A Fc Chimer (Catalog # 10203-CL) is immobilized at 5 μg/mL, 100 μL/well, the concentration of Recombinant Human DEC-205/CD205 Fc Chimera (Catalog # 10205-DE) that produces 50% of the optimal binding response is 4-24 μg/mL.
Scientific Data Images for Recombinant Human CD302/CLEC13A Fc Chimera Protein, CF
Recombinant Human CD302/CLEC13A Fc Chimera Protein SDS-PAGE
2 μg/lane of Recombinant Human CD302/CLEC13A Fc Chimera was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing bands at 51-59 kDa and 100-120 kDa, respectively.Formulation, Preparation and Storage
10203-CL
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 500 μg/mL in PBS. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: CD302/CLEC13A
References
- Kato, M. et al. (2007) J. Immunol. 179:6052.
- Lo, T-H. et al. (2016) J. Immunol. 197:885.
- Pospisilova, E. et al. (2016) Biomol. NMR. Assign. 10:189.
- Kato, M. et al. (2003) J Biol Chem. 278:34035.
- Butler, M. et al. (2017) J. Immunol. 120:362.
- Lo, T-H. et al. (2019) PLoS One. 14:e0216368.
Alternate Names
BIMLEC, CD302, CLEC13A, DCL1
Gene Symbol
CD302
UniProt
Additional CD302/CLEC13A Products
Product Documents for Recombinant Human CD302/CLEC13A Fc Chimera Protein, CF
Product Specific Notices for Recombinant Human CD302/CLEC13A Fc Chimera Protein, CF
For research use only
Loading...
Loading...
Loading...
Loading...