Recombinant Guinea Pig Asparaginase His-tag Protein, CF
R&D Systems, part of Bio-Techne | Catalog # 10238-AS

Key Product Details
- R&D Systems E. coli-derived Recombinant Guinea Pig Asparaginase His-tag Protein (10238-AS)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Product Specifications
Source
E. coli-derived guinea pig Asparaginase protein
Ala2-Ile565
with an N-terminal Met and 6-His tag
Ala2-Ile565
with an N-terminal Met and 6-His tag
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<1.0 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Met
Predicted Molecular Mass
62 kDa
SDS-PAGE
57 kDa, under reducing conditions
Activity
Measured by its ability to convert asparagine to aspartic acid.
The specific activity is >2700 pmol/min/μg, as measured under the described conditions.
The specific activity is >2700 pmol/min/μg, as measured under the described conditions.
Scientific Data Images for Recombinant Guinea Pig Asparaginase His-tag Protein, CF
Recombinant Guinea Pig Asparaginase His-tag Protein Binding Activity
Recombinant Guinea Pig ASPG His-tag (Catalog # 10238-AS) is measured by its ability to convert asparagine to aspartic acid.Recombinant Guinea Pig Asparaginase His-tag Protein SDS-PAGE
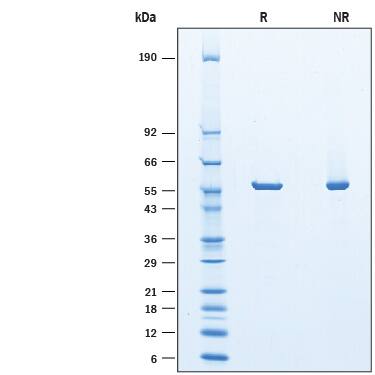
2 μg/lane of Recombinant Guinea Pig Asparaginase His-tag was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing a band under reducing conditions at 57 kDa.Formulation, Preparation and Storage
10238-AS
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, NaCl and TCEP. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Asparaginase
References
- Vimal, A. and A. Kumar (2017) Biotechnol. Genet. Eng. Rev. 33:40.
- Schalk, A. M. et al. (2014) J. Biol. Chem. 289:33175.
- Kidd, J. G. (1953) J. Exp. Med. 98:565.
- Broome, J. D. (1963) J. Exp. Med. 118:99.
- Neuman, R. E. and T. A. McCoy (1956) Science 124:124.
- Haley, E. E. et al. (1961) Cancer Res. 21:532.
- Su, N. et al. (2008) Pediatr. Blood Cancer. 50:274.
- Salzer, W. L. et al. (2014) Ann. N.Y. Acad. Sci. 1329:81.
- Muller, H. J. and J. Boos (1998) Crit. Rev. Oncol. Hematol. 28:97.
- Chan, W. K. et al. (2014) Blood 123:3596.
- Dellinger, C. T. and T. D. Miale (1976) Cancer 38:1843.
- Belviso, S. et al. (2017) PLoS One. 12:e0178174.
- Karamitros, C. S. and M. Konrad (2014) J. Biol. Chem. 289:12962.
- Srikhanta, Y. N. et al. (2013) Biochem. Biophys. Res. Commun. 436:362.
Long Name
Asparaginase
Alternate Names
ASPG, C14orf76
Gene Symbol
ASPG
UniProt
Additional Asparaginase Products
Product Documents for Recombinant Guinea Pig Asparaginase His-tag Protein, CF
Product Specific Notices for Recombinant Guinea Pig Asparaginase His-tag Protein, CF
For research use only
Loading...
Loading...
Loading...