Sphingomyelin Assay Kit (Colorimetric)
Novus Biologicals, part of Bio-Techne | Catalog # KA1373

Key Product Details
Sample Type & Volume Required Per Well
Serum or Plasma (10 uL/well. All samples should be assayed in duplicate at minimum.)
Assay Range
5 to 50 mg/dL
Product Specifications
Assay Type
Colorimetric
Kit Type
Assay Kit (Colorimetric)
Description
Quality control test: Standard curve Sphingomyelin Assay Kit provides a method for quantifying sphingomyelin in plasma or serum.
Scientific Data Images for Sphingomyelin Assay Kit (Colorimetric)
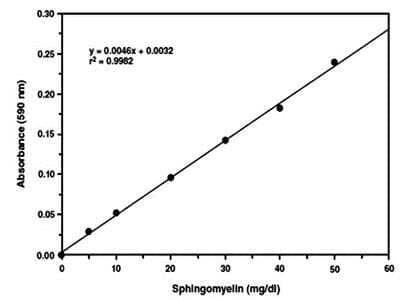
Sphingomyelin Assay Kit (Colorimetric) [KA1373] - The standard curve is for the purpose of illustration only and should not be used to calculate unknowns. A standard curve should be generated each time the assay is performed.
Kit Contents for Sphingomyelin Assay Kit (Colorimetric)
- 96-Well Cover Sheet
- 96-Well Plate (Colorimetric Assay)
- SM Alkaline Phosphatase
- SM Buffer (10X)
- SM Color Detector
- SM Detergent Solution
- SM Enzyme Mixture
- Sphingomyelin Standard
- Sphingomyelinase
Preparation and Storage
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Store at 4°C.
Background: Sphingomyelin
Additional Sphingomyelin Products
Product Documents for Sphingomyelin Assay Kit (Colorimetric)
Product Specific Notices for Sphingomyelin Assay Kit (Colorimetric)
This product is produced by and distributed for Abnova, a company based in Taiwan.
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Kits are guaranteed for 6 months from date of receipt.
Loading...
Loading...
Loading...