Human Blood B Booster Kit
Novus Biologicals, part of Bio-Techne | Catalog # DDXK-HuBBB

Discontinued Product
DDXK-HuBBB has been discontinued.
View all Human Blood B Booster products.
Key Product Details
Species
Human
Applications
Functional Assay
Kit Type
Kit
Product Summary for Human Blood B Booster Kit
Dendritics has developed the HuBBB(R) kit as an improvement of the above approaches, in order to target the largest B cell population from peripheral blood, thus offering the opportunity to pursue studies on immortalized B cells representative of the overall peripheral B repertoire. Under the best conditions, the kit allows the immortalization of up to 20% of the starting B cell population. The HuBBB(R) kit was packaged so as to make it very easy to handle for the scientists ; it contains all the reagents needed to obtain from 5.104 to 2.105 independent B cell clones from 107 peripheral blood mononuclear cells using ten 96 wells culture plates. The resulting cells are available for a wide range of studies including genomic, transcriptomic and proteomic fields.
Intended usage: -Production of human monoclonal antibodies for research use. -Drug discovery/development: development of fully human antibodies for use in in vitro studies. -Evaluation of biotherapeutics agents or candidate vacines on immunoglobulin secretion. -Measurment of auto/allo-antibodies against specific biological targets like cytokines. Successful Proofs of Concepts: -Fully human monoclonal antibodies were made against several targets including HuMAB anti-HPA5b, -IL1a, -BetV1, -Derp1 -Neutralizing HumAB anti-Chik -Hu and canMab anti-Leptospira
Product Specifications
Application Notes
Intended use: Immortalization of human B lymphocytes from peripheral blood samples
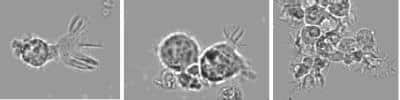
Scientific Data Images for Human Blood B Booster Kit
B Cell Stimulation: Human Blood B Booster Kit [DDXK-HuBBB] - Microscopic observation during the time course of the culture.
Kit Contents for Human Blood B Booster Kit
- Booster reagent
- Vial #1 - support 1
- Vial #2 - mAb1
- Vial #3 - mAb2
- Vial #4 - EBV1
- Vial #5 - support2
- Vial #6 - mAb3
- Vial #7 - EBV2
Formulation, Preparation, and Storage
Concentration
Concentration is not relevant for this product. Please see the protocols for proper use of this product.
Shipping
The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below.
Storage
Storage of components varies. See protocol for specific instructions.
Background: Human Blood B Booster
Alternate Names
Blood B Booster, Blood B Booster kit, human B cell immortalization Human Blood B Booster, Human B cell immortalization kit, Human Blood B Booster, Human Blood B Booster kit
Additional Human Blood B Booster Products
Product Documents for Human Blood B Booster Kit
Product Specific Notices for Human Blood B Booster Kit
This product is manufactured by Eurobio Scientific (formerly Dendritics) and distributed by Novus Biologicals.
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Kits are guaranteed for 6 months from date of receipt.
Loading...
Loading...
Loading...