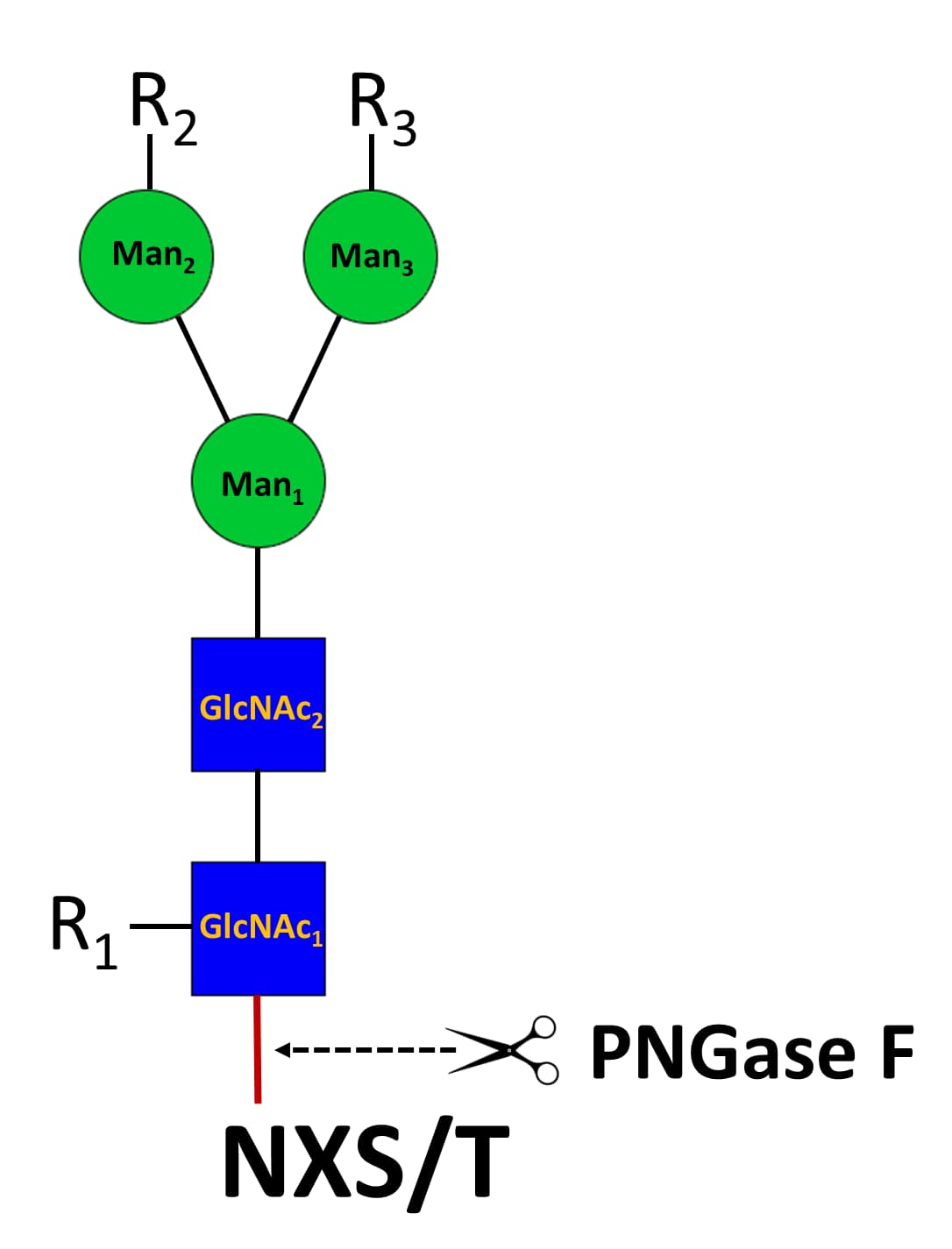
PNGase F N-glycan Releasing Kit
R&D Systems, part of Bio-Techne | Catalog # EA006

Key Product Details
| Key Benefits: |
Learn more about Fluorescent Glycan Labeling and Detection |
Assay Procedure
ASSAY PROTOCOL
Denaturing Assay Protocol
Reaction volumes can be increased or decreased proportionally. It is recommended to start with concentrated samples so that less sample volume is required, therefore less denaturing buffer will be applied to final reactions to minimize the negative effects of SDS on PNGase F. PNGase F can be directly used or diluted in 1X PNGase F Assay Buffer before usage. A titration on PNGase F maybe performed to determine amount of enzyme is needed for a specific sample protein. For negative controls, replace PNGase F with 1X Assay Buffer. Make sure to mix well all components in each step.
Reagent Preparation
Prepare 1X Assay Buffer by diluting the 10X PNGase F Assay Buffer with deionized or distilled water.
- Sample Preparation
- In a centrifuge tube add the following items stepwise, 10 µL Sample Protein or Cy5-RBD and 1 µL Denaturing Buffer (10X).
- Heat at 95°C for 5 minutes and then chill on ice. Briefly centrifuge condensation.
- Add 1 µL Renaturing Buffer (10X)
Note: The sample protein may be diluted with 1X PNGase F Assay Buffer to a desired concentration here.
- PNGase F Digestion
- Add 5 µL of Denatured Sample Protein or Cy5-RBD, 14 µL of 1X Assay Buffer, and 1 µL PNGase F to a new centrifuge tube.
- Incubate at 37 °C for 60 minutes to overnight.
- Add 4 µL of 6X SDS Gel Loading Buffer to the tube.
- SDS-Gel Separation and Imaging
- Load half of the final digestion volume to an SDS Gel.
- Run the gel until the dye front reaches the end of the gel.
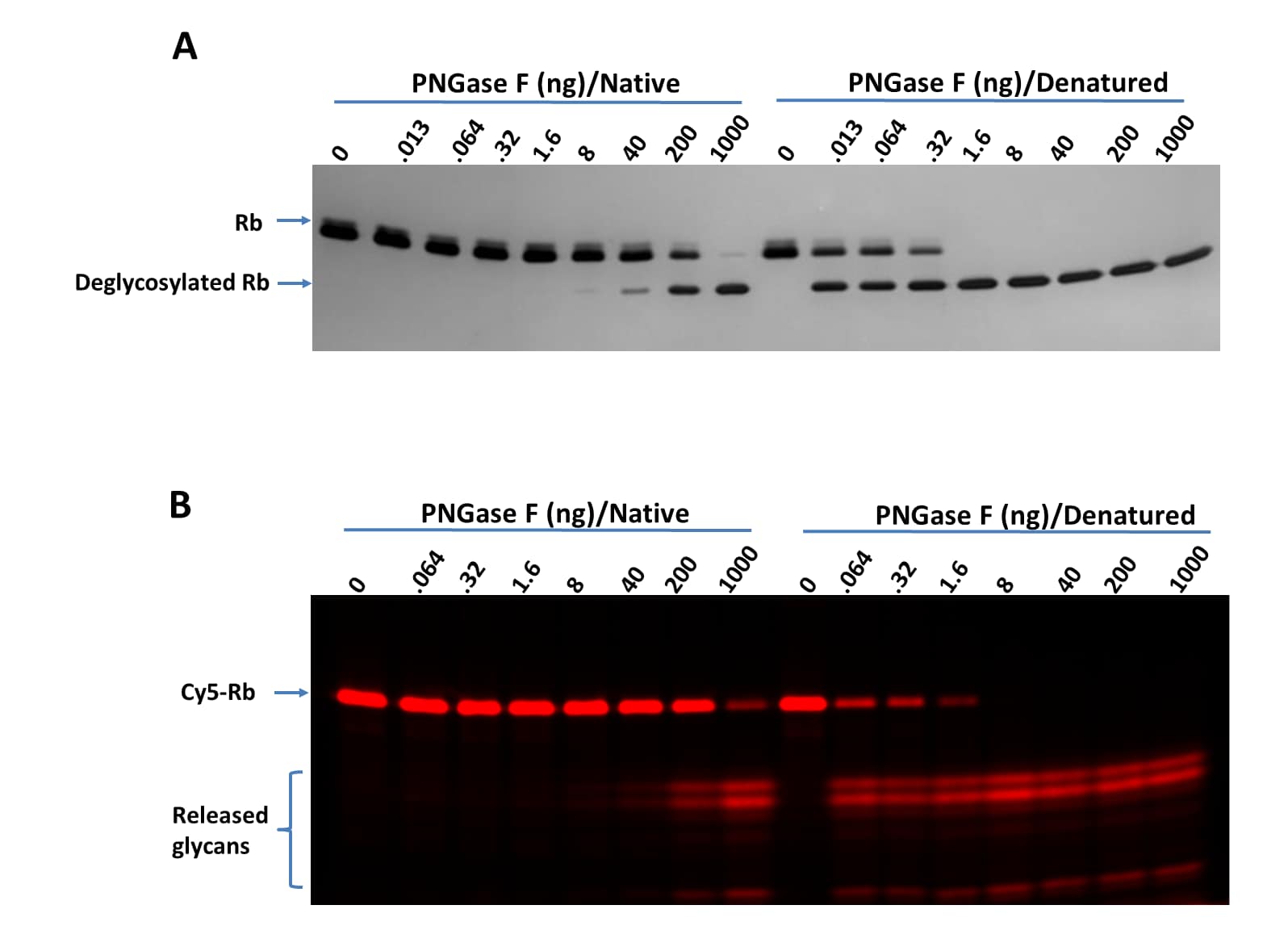
- Perform fluorescent imaging first if Cy5-RBD is used as a control.
- Perform regular protein imaging via Silver, TCE or Coomassie® Blue staining.
NATIVE ASSAY PROTOCOL
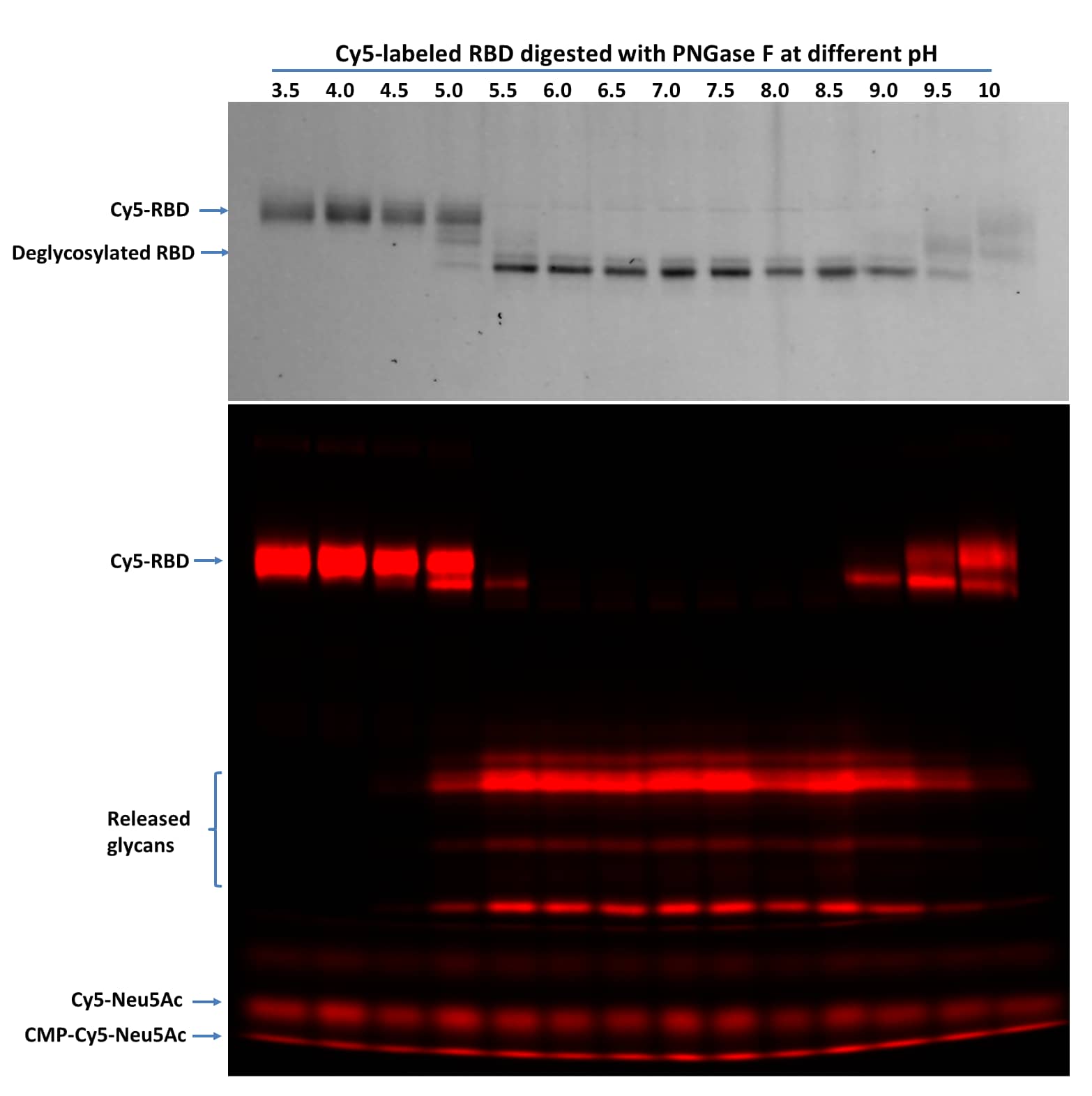
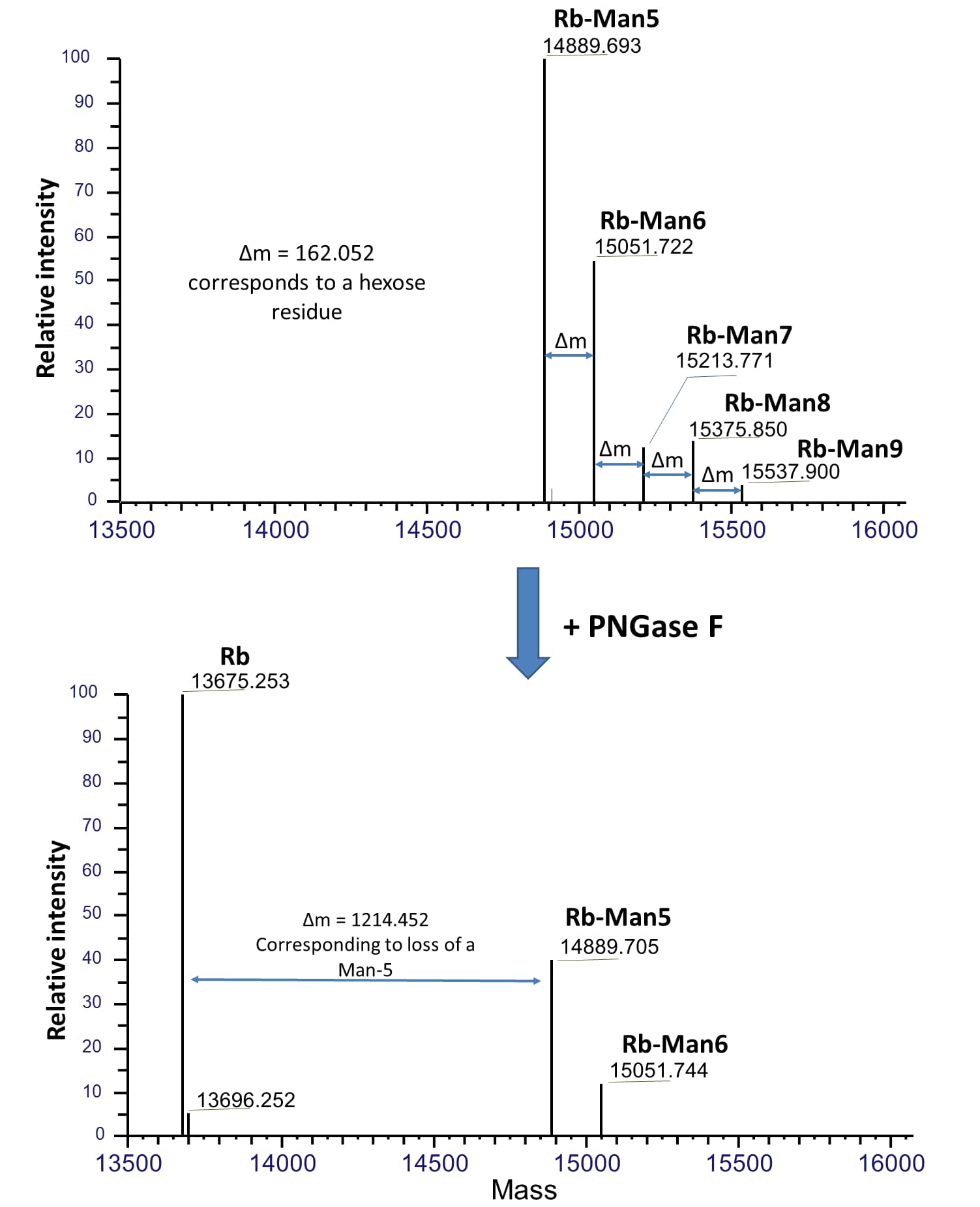
Native assay protocol is specifically for samples that are intended for mass spec analysis. Reaction volumes can be increased proportionally. Proteins under native conditions are much more resistant to PNGase F digestion. To completely deglycosylate proteins under native conditions, high concentration PNGase F and longer incubation time are recommended.
Customer Reviews for PNGase F N-glycan Releasing Kit
There are currently no reviews for this product. Be the first to review PNGase F N-glycan Releasing Kit and earn rewards!
Have you used PNGase F N-glycan Releasing Kit?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review