Recombinant Human Nectin-2/CD112 Protein
R&D Systems, part of Bio-Techne | Catalog # 2229-N2

Key Product Details
- R&D Systems NS0-derived Recombinant Human Nectin-2/CD112 Protein (2229-N2)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession #
Structure / Form
Conjugate
Applications
Product Specifications
Source
Gln32-Leu360, with a C-terminal 6-His tag
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
Scientific Data Images for Recombinant Human Nectin-2/CD112 Protein
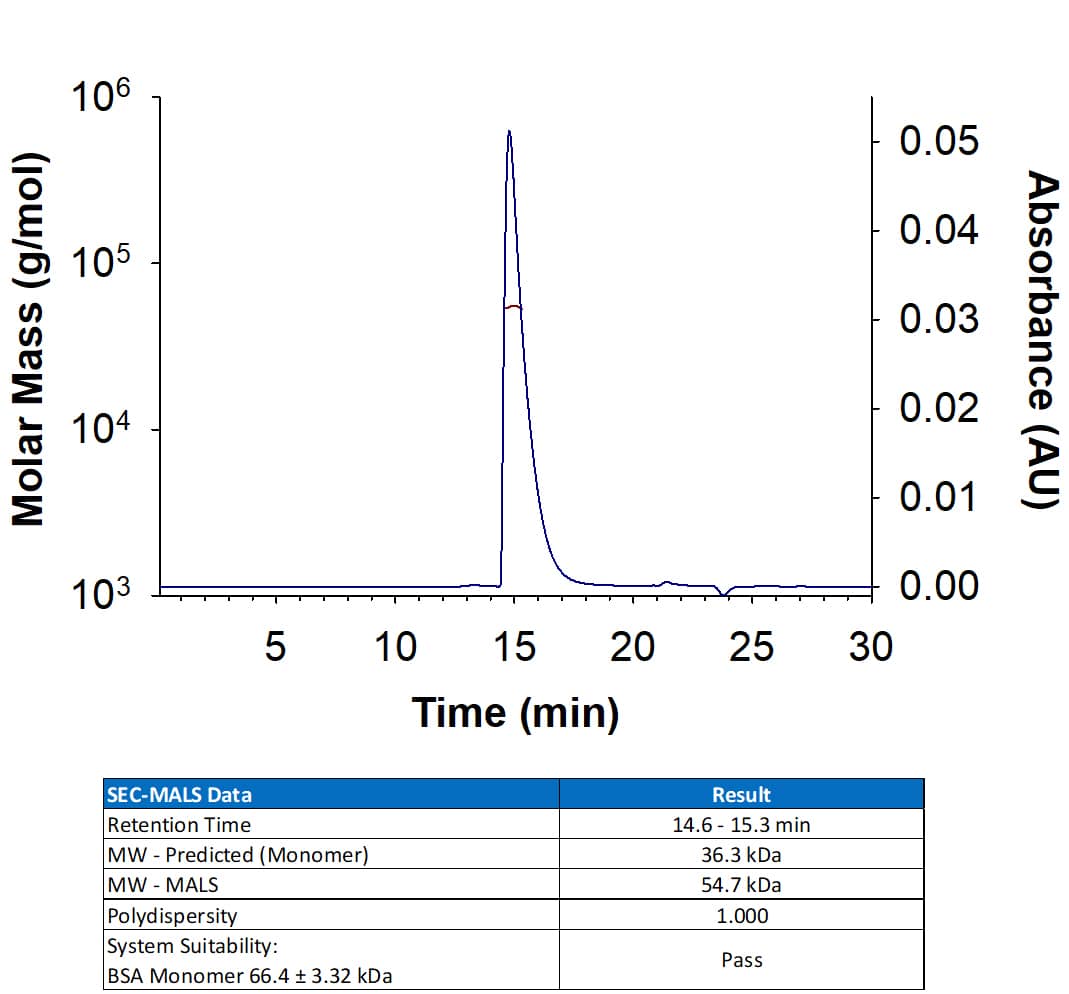
Recombinant Human Nectin-2/CD112 Protein SEC-MALS.
Recombinant Human Nectin-2/CD112 Protein (Catalog # 2229-N2) has a molecular weight (MW) of 52-57 kDa as analyzed by SEC-MALS, suggesting that this protein is a monomer. MW may differ from predicted MW due to post-translational modifications (PTMs) present (i.e. Glycosylation).Formulation, Preparation and Storage
Carrier Free
What does CF mean?CF stands for Carrier Free (CF). We typically add Bovine Serum Albumin (BSA) as a carrier protein to our recombinant proteins. Adding a carrier protein enhances protein stability, increases shelf-life, and allows the recombinant protein to be stored at a more dilute concentration. The carrier free version does not contain BSA.
What formulation is right for me?In general, we advise purchasing the recombinant protein with BSA for use in cell or tissue culture, or as an ELISA standard. In contrast, the carrier free protein is recommended for applications, in which the presence of BSA could interfere.
Carrier: 2229-N2
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile PBS containing at least 0.1% human or bovine serum albumin. |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Carrier Free: 2229-N2/CF
| Formulation | Supplied as a 0.2 μm filtered solution in PBS. |
| Reconstitution | It is recommended that sterile PBS be added to the vial to prepare a working stock solution of no less than 50 μg/mL. The carrier-free protein should be used immediately upon reconstitution to avoid losses in activity due to non-specific binding to the inside surface of the vial. For long term storage as a dilute solution, a carrier protein (e.g. 0.1% HSA or BSA) should be added to the vial. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Nectin-2/CD112
Nectins are a small family of Ca++-independent immunoglobulin (Ig)-like cell adhesion molecules (CAMs) that organize intercellular junctions (1). The nectin family has at least four members (nectin-1-4), all of which show alternate splicing (except for Nectin-4), a transmembrane (TM) region (except for Nectin-1 gamma), and three extracellular Ig-domains. Nectins are highly homologous to the human receptor for poliovirus, and as such have been alternately named poliovirus receptor-related proteins. They do not, however, appear to bind poliovirus (1). Nectin-2 is a 60 or 65 kDa type I TM glycoprotein that is found on a variety of cell types (2, 3). It has two splice forms (4, 5). Nectin-2 delta is a 65 kDa long form and is synthesized as a 538 amino acid precursor. It contains a 31 amino acid (aa) signal sequence, a 329 aa extracellular region, a 21 aa TM segment, and a 157 aa cytoplasmic domain. The extracellular region contains one N-terminal 85 aa V-type Ig domain and
two 45-55 aa C2-type Ig domains. The V-domain is believed to mediate nectin binding to its ligands (6). The short, 60 kDa isoform of Nectin-2 (Nectin-2 alpha) has the same signal sequence and extracellular domain as nectin-2 delta, but differs in the TM and cytoplasmic region (4, 5). In this case, the cytoplasmic tail is only 94 aa in length. The human extracellular region shows 72% aa sequence identity with the equivalent region in mouse. Nectin-2 is known to bind the pseudorabies virus, and herpes simplex virus-2 (HSV-2), but not HSV-1. It does not bind poliovirus. As a cell adhesion molecule, Nectin-2 will form cis-homodimers (same cell), followed by trans-dimers (across cells). Nectin-2 will not cis-dimerize with other nectins, but will cis-dimerize with its two splice forms. Notably, a Nectin-2 cis-dimer on one cell will heterodimerize with a Nectin-3 cis-dimer on another cell (1). Nectin-2 is found concentrated in adherens junctions, and exists on neurons, endothelial cells, epithelial cells and fibroblasts.
References
- Takai, Y. and H. Nakanishi, 2003, J. Cell Sci. 116:17.
- Bottino, C. et al. (2003) J. Exp. Med. 198:557.
- Pende, D. et al. (2005) Mol. Immunol. 42:463.
- Eberle, F. et al. (1995) Gene 159:267.
- Warner, M.S. et al. (1998) Virology 246:179.
- Struyf, F. et al. (2002) J. Virol. 76:12940.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional Nectin-2/CD112 Products
Product Documents for Recombinant Human Nectin-2/CD112 Protein
Product Specific Notices for Recombinant Human Nectin-2/CD112 Protein
For research use only