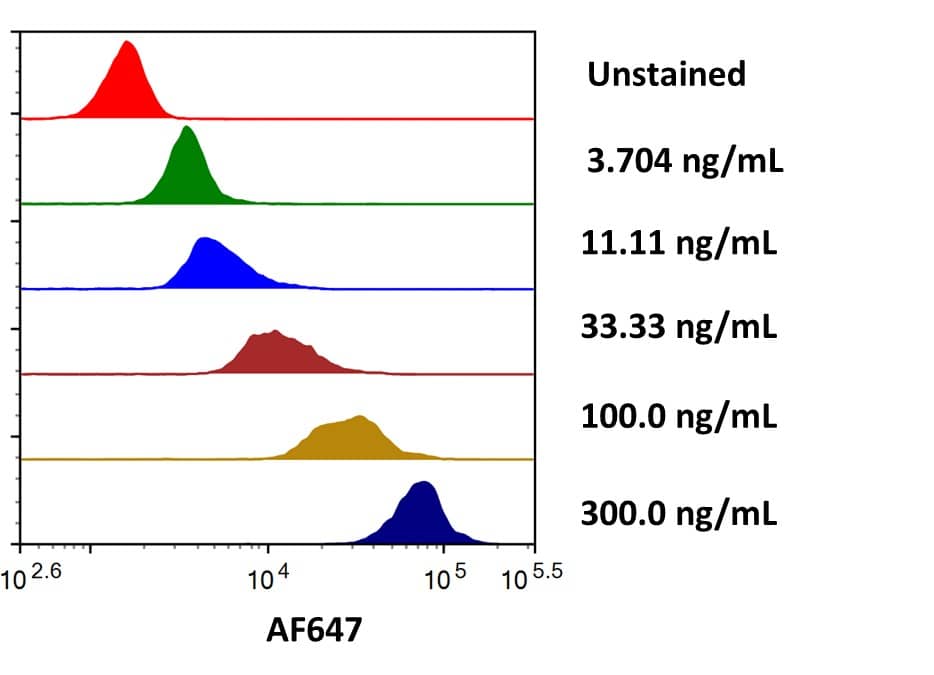
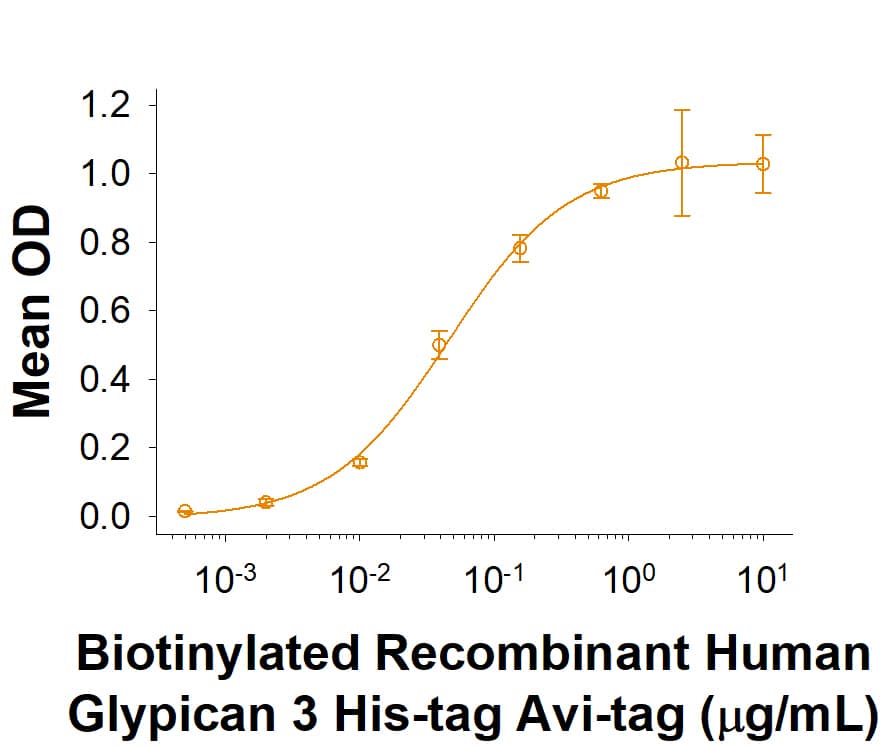
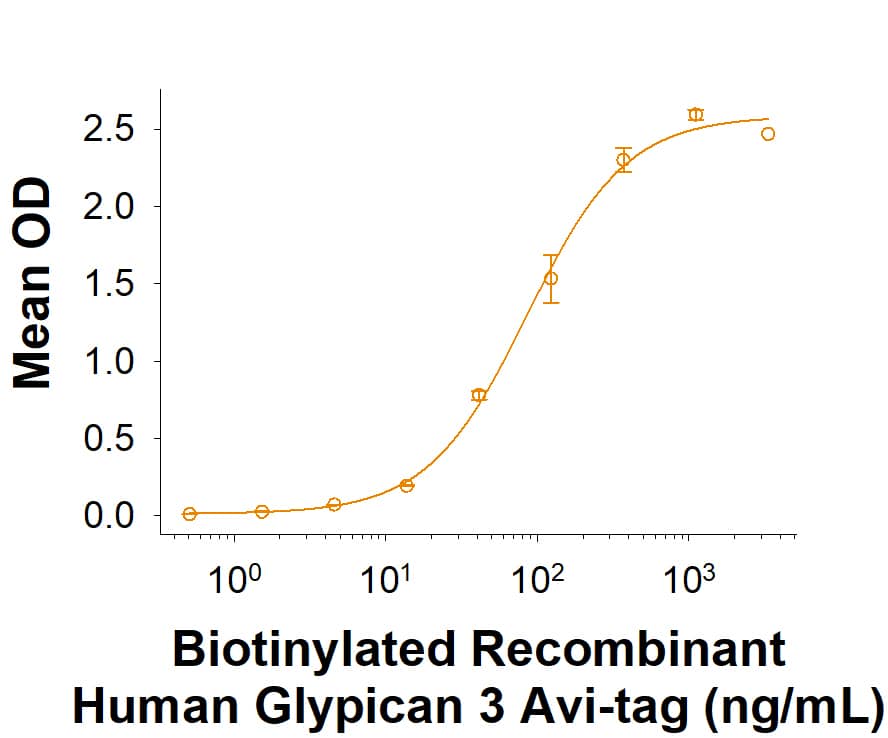
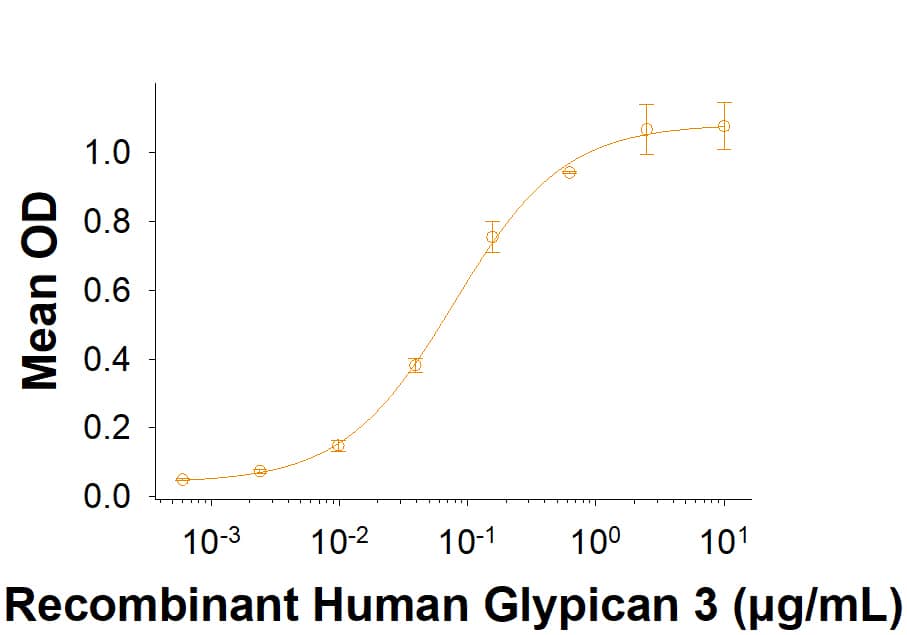
Glypican 3: Proteins and Enzymes
The glypicans (GPC) constitute a family of glycosylphosphatidylinositol (GPI)-anchored, heparan sulfate proteoglycans. Six members of this family have been identified in mammals (GPC1-GPC6). All glypican core proteins contain an N-terminal signal peptide, a large globular cysteine-rich domain (CRD) with 14 invariant cysteine residues, a stalk-like region containing the heparan sulfate attachment sites, and a C-terminal GPI attachment site.
Mutations in Glypican 3 cause a rare disorder in humans, Simpson-Golabi-Behmel Syndrome, which is characterized by pre- and postnatal overgrowth of multiple tissues and organs and an increased risk for developing embryonic tumors. These features are also present in the mouse knock-out of Glypican 3 indicating that Glypican 3 regulates cell survival and inhibits cell proliferation during development. Glypican 3 has been implicated in regulating many different signaling pathways including: IGF, FGF, BMP and Wnt. An endoproteolytic processing of Glypican 3 by proprotein convertases is required for the modulation of Wnt signaling. Direct interaction of Glypican 3 with FGF-basic has been observed and is mediated by the heparan sulfate chains.
9 results for "Glypican 3 Proteins and Enzymes" in Products
9 results for "Glypican 3 Proteins and Enzymes" in Products
Glypican 3: Proteins and Enzymes
The glypicans (GPC) constitute a family of glycosylphosphatidylinositol (GPI)-anchored, heparan sulfate proteoglycans. Six members of this family have been identified in mammals (GPC1-GPC6). All glypican core proteins contain an N-terminal signal peptide, a large globular cysteine-rich domain (CRD) with 14 invariant cysteine residues, a stalk-like region containing the heparan sulfate attachment sites, and a C-terminal GPI attachment site.
Mutations in Glypican 3 cause a rare disorder in humans, Simpson-Golabi-Behmel Syndrome, which is characterized by pre- and postnatal overgrowth of multiple tissues and organs and an increased risk for developing embryonic tumors. These features are also present in the mouse knock-out of Glypican 3 indicating that Glypican 3 regulates cell survival and inhibits cell proliferation during development. Glypican 3 has been implicated in regulating many different signaling pathways including: IGF, FGF, BMP and Wnt. An endoproteolytic processing of Glypican 3 by proprotein convertases is required for the modulation of Wnt signaling. Direct interaction of Glypican 3 with FGF-basic has been observed and is mediated by the heparan sulfate chains.
| Source: | NS0 |
| Accession #: | P51654.1 |
| Applications: | Bind |
| Source: | NS0 |
| Accession #: | P51654.1 |
| Applications: | BA |
| Source: | CHO |
| Accession #: | P51654.1 |
| Applications: | BA |
| Source: | NS0 |
| Accession #: | AAH36126 |
| Applications: | BA |
| Source: | HEK293 |
| Accession #: | XP_005594665.1 |
| Applications: | BA |
| Source: | CHO |
| Applications: | BA |
| Source: | HEK293 |
| Accession #: | P51654.1 |
| Applications: | BA |
| Source: | NS0 |
| Accession #: | P51654.1 |
| Applications: | BA |