Recombinant Human IL-2 (Mammalian-expressed) Protein, CF
R&D Systems, part of Bio-Techne | Catalog # 10453-IL

Key Product Details
- R&D Systems CHO-derived Recombinant Human IL-2 (Mammalian-expressed) Protein (10453-IL)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession #
Structure / Form
Conjugate
Applications
Product Specifications
Source
Ala21-Thr153
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The ED50 for this effect is 0.05-0.3 ng/mL.
Reviewed Applications
Read 1 review rated 5 using 10453-IL in the following applications:
Scientific Data Images for Recombinant Human IL-2 (Mammalian-expressed) Protein, CF
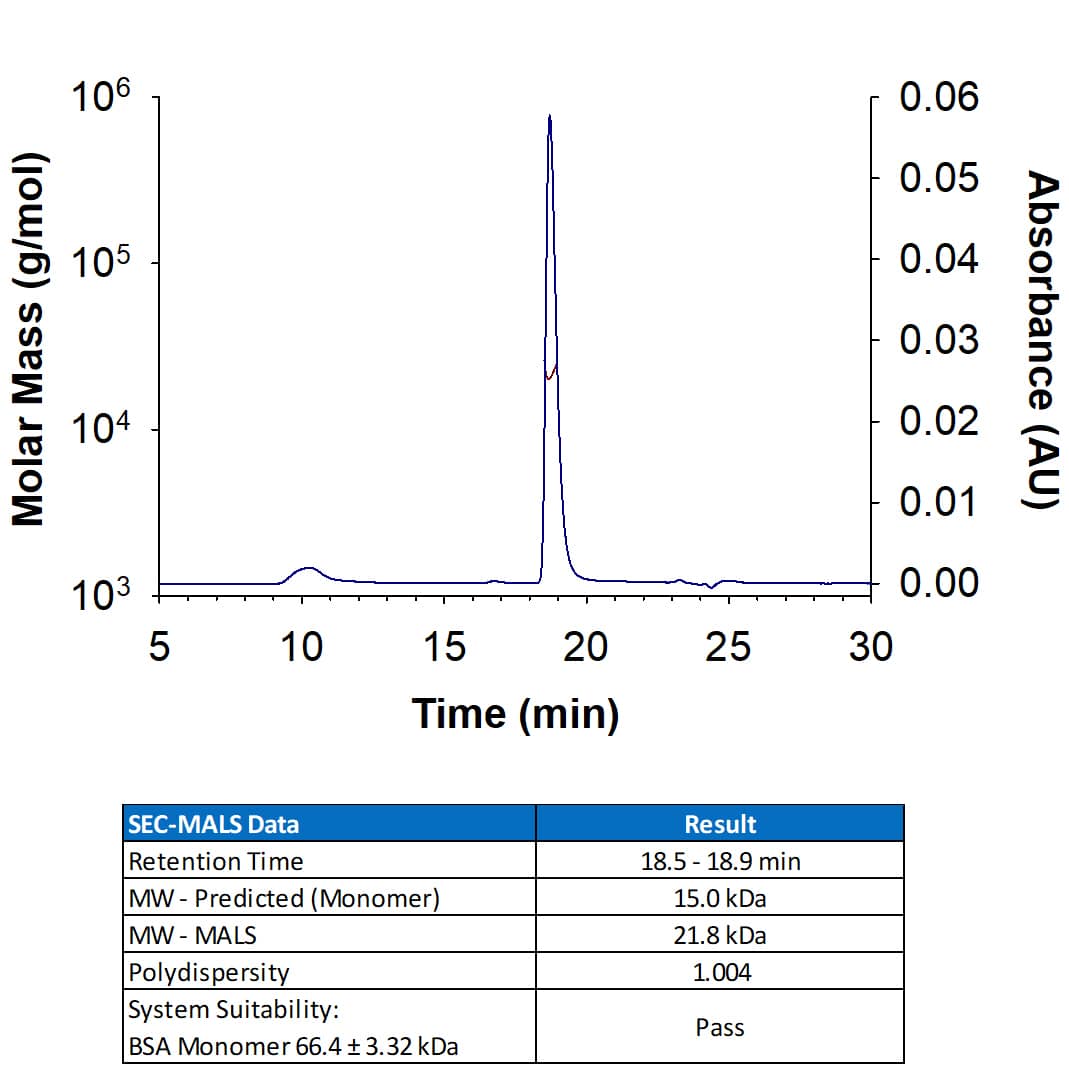
Recombinant Human IL‑2 (Mammalian-expressed) Protein SEC-MALS.
Recombinant Human IL‑2 (Mammalian-expressed) Protein (Catalog # 10453-IL) has a molecular weight (MW) of 21-23 kDa as analyzed by SEC-MALS, suggesting that this protein is a monomer. MW may differ from predicted MW due to post-translational modifications (PTMs) present (i.e. Glycosylation).Recombinant Human IL-2 (Mammalian-expressed) Protein Bioactivity
Recombinant Human IL-2 (Mammalian-expressed) Protein (10453-IL) stimulates cell proliferation of the CTLL-2 mouse cytotoxic T cell line. The ED50 for this effect is 0.05-0.3 ng/mL. Mammalian-expressed Recombinant Human IL-2 has similar activity to E.coli-derived Recombinant Human IL-2 (202-IL).Recombinant Human IL-2 (Mammalian-expressed) Protein SDS-PAGE
2 μg/lane of Recombinant Human IL-2 (Mammalian-expressed) Protein (10453-IL) was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing bands at 14-18 kDa.Formulation, Preparation and Storage
10453-IL
| Formulation | Lyophilized from a 0.2 μm filtered solution in Acetonitrile and TFA. |
| Reconstitution | Reconstitute at 100-500 μg/mL in sterile 100 mM Acetic Acid |
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: IL-2
Recombinant IL-2 is widely used in cell culture for the expansion of T cells. IL-2 is expressed by CD4+ and CD8+ T cells, gamma delta T cells, B cells, dendritic cells, and eosinophils (1-3). Mature human IL-2 shares 56% and 66% amino acid (aa) sequence identity with mouse and rat IL-2, respectively. Human and mouse IL-2 exhibit cross-species activity (4). The receptor for IL-2 consists of three subunits that are present on the cell surface in varying preformed complexes (5-7). The 55 kDa IL-2 R alpha is specific for IL-2 and binds with low affinity. The 75 kDa IL-2 R beta, which is also a component of the IL-15 receptor, binds IL-2 with intermediate affinity. The 64 kDa common gamma chain gammac/IL-2 R gamma, which is shared with the receptors for IL-4, -7, -9, -15, and -21, does not independently interact with IL-2. Upon ligand binding, signal transduction is performed by both IL-2 R beta and gammac.
IL-2 is best known for its autocrine and paracrine activity on T cells. It drives resting T cells to proliferate and induces IL-2 and IL-2 R alpha synthesis (1, 2). It contributes to T cell homeostasis by promoting the Fas-induced death of naïve CD4+ T cells but not activated CD4+ memory lymphocytes (8). IL-2 plays a central role in the expansion and maintenance of regulatory T cells, although it inhibits the development of Th17 polarized cells (9-11). Thus, IL-2 may be a key cytokine in the natural suppression of autoimmunity (12, 13).
IL-2 expression and concentration can have either immunostimulatory effects at high doses or immunosuppressive effects at low doses due to its preferential binding to different receptor subunits expressed by various immune cell types. This has led to the generation of recombinant IL-2 variants aimed at modifying IL-2 receptor binding for increased antitumor efficacy (14, 15). These variants are typically used in combination with immune checkpoint inhibitors instead of as a monotherapy (14). IL-2 can be genetically engineered to express in NK cells for CAR T cell therapies, and in combination with other cytokines like IL-15, can increase cell viability and proliferation (16). In addition to adoptive cell transfer and checkpoint blockade inhibitors, cancer vaccines that boost immune responses have been combined with IL-2 treatment with promising results in recent studies (15).
In cell culture, IL-2 is a frequently used cytokine for the proliferation, differentiation, and increased antibody secretion of B cells as they transform into plasma cells in vitro (17). IL-2 is also a classically used cytokine for the expansion of NK cells, early differentiated T cells and effector memory Treg cells for adoptive cell transfer cancer immunotherapy (16, 18). GMP IL-2 is a commonly used supplement for the expansion of these cell types for cellular therapies.
References
- Ma, A. et al. (2006) Annu. Rev. Immunol. 24:657.
- Gaffen, S.L. and K.D. Liu (2004) Cytokine 28:109.
- Taniguchi, T. et al. (1983) Nature 302:305.
- Mosmann, T.R. et al. (1987) J. Immunol. 138:1813.
- Liparoto, S.F. et al. (2002) Biochemistry 41:2543.
- Wang, X. et al. (2005) Science 310:1159.
- Bodnar, A. et al. (2008) Immunol. Lett. 116:117.
- Jaleco, S. et al. (2003) J. Immunol. 171:61.
- Malek, T.R. (2003) J. Leukoc. Biol. 74:961.
- Laurence, A. et al. (2007) Immunity 26:371.
- Kryczek, I. et al. (2007) J. Immunol. 178:6730.
- Afzali, B. et al. (2007) Clin. Exp. Immunol. 148:32.
- Fehervari, Z.et al. (2006) Trends Immunol. 27:109.
- Xue, D. et al. (2021) Antib Ther. 4:123.
- Wolfarth, A.A. et al. (2022) Immune Netw. 22:e5.
- Koehl, U. et al. (2015) Oncoimmunology. 5:e1115178.
- Marsman, C. et al. (2022) Front. In Immunol. 13:815449.
- Chamucero-Millares, J.A. et al. (2021) Cellular Immunol. 360:104257.
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
UniProt
Additional IL-2 Products
Product Documents for Recombinant Human IL-2 (Mammalian-expressed) Protein, CF
Product Specific Notices for Recombinant Human IL-2 (Mammalian-expressed) Protein, CF
For research use only