Recombinant Virus Dengue Virus 1 VLP Protein
Novus Biologicals, part of Bio-Techne | Catalog # NBP3-14811

![Western Blot: Recombinant Virus Dengue Virus 1 VLP Protein [NBP3-14811] Western Blot: Recombinant Virus Dengue Virus 1 VLP Protein [NBP3-14811]](https://resources.bio-techne.com/images/products/Dengue-Virus-1-VLP-Recombinant-Protein-Antigen-Western-Blot-NBP3-14811-img0002.jpg)
Discontinued Product
NBP3-14811 has been discontinued.
View all Dengue Virus 1 VLP products.
Key Product Details
Source
HEK293
Conjugate
Unconjugated
Applications
Western Blot, Microscopy
Product Specifications
Description
Recombinant Dengue Virus serotype 1 virus-like particle recombinant antigen consisting of Envelope, pre-Membrane and Membrane protein (NCBI Accession Number: ABO45246.1). The C-terminal 20% of the dengue Envelope protein was replaced by the corresponding Japanese Encephalitis Virus sequence (all internal/transmembrane).
Storage: Avoid excessive mixing or shocking to prevent aggregation. Long term storage above -80C may result in aggregate formation.
Concentration: 0.1-0.6mg/ml
Storage: Avoid excessive mixing or shocking to prevent aggregation. Long term storage above -80C may result in aggregate formation.
Concentration: 0.1-0.6mg/ml
Purity
>95% pure by SDS-PAGE
Protein / Peptide Type
Recombinant Protein
Scientific Data Images for Recombinant Virus Dengue Virus 1 VLP Protein
Western Blot: Recombinant Virus Dengue Virus 1 VLP Protein [NBP3-14811]
Western Blot: Dengue Virus 1 VLP Recombinant Protein Antigen [NBP3-14811] - Non-reducing SDS-PAGE gel showing purified Dengue virus serotype 1 virus-like particles; envelope protein (E) at approx. 55kDa, and prM protein running at approx. 18kDa.
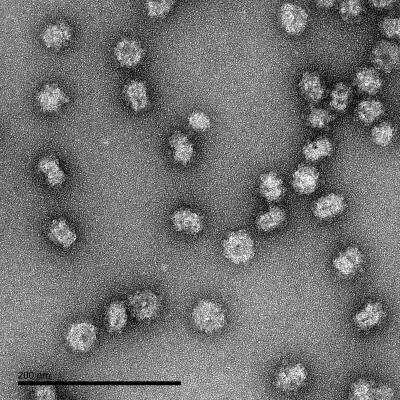
Electron Microscopy: Dengue Virus 1 VLP Recombinant Protein Antigen [NBP3-14811] - Negative staining electron microscopy of DENV1 virus-like particles (VLPs).
Formulation, Preparation and Storage
NBP3-14811
| Preparation Method | Recombinant Dengue Virus serotype 1 virus-like particle recombinant antigen consisting of Envelope, pre-Membrane and Membrane protein produced in HEK293 human cells. The C-terminal 20% of the dengue Envelope protein was replaced by the corresponding Japanese Encephalitis virus sequence. Tested for stability, concentrated and purified by a series of ultracentrifugation and chromatographical methods which result in VLPs of exceptional quality and purity. |
| Formulation | 10mM sodium phosphate, 20mM sodium citrate, 154mM sodium chloride, pH7.4 |
| Preservative | No Preservative |
| Concentration | Please see the vial label for concentration. If unlisted please contact technical services. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Store at 4C short term. Aliquot and store at -80C long term. Avoid freeze-thaw cycles. |
Background: Dengue Virus 1 VLP
Dengue virus entry into host cells occurs via receptor-mediated endocytosis by receptor molecules including the mannose receptor, heparan sulfate, glycosaminoglycans, and DC-SIGN (1,3). Following attachment, the virus is endocytosed in clathrin-coated vesicles (1,3). Following internalization, clathrin disassembles and endosomal processing occurs, allowing viral fusion, disassembly, and release of viral RNA (1,3). This release results in viral translation and replication, virus assembly and maturation, and eventual exocytosis of the mature virus (1,3). Infection can result in a wide range of clinical symptoms including mild disease such as Dengue fever which is characterized by fever, headache, joint pain, rash, and retro-orbital pain, or severe, life-threatening conditions like Dengue hemorrhagic fever or Dengue shock syndrome which involves vascular permeability and leakage (1-3). Host immune response against infection includes innate immune response via interferon secretion and pro-inflammatory cytokine production, as well as adaptive immune response involving cellular and humoral components like T cell activation and B-cell mediated antibody production (1-3). As far as treatment for Dengue virus infection, there no commercial antiviral agents, though some anti-pyretics and certain phenolic compounds do show promise in treating infection (1-3). However, Resveratol, an antiviral for other Flavivirus, has been shown to directly attack the Dengue virus genome (1). While more work needs to be done, there are some live-attenuated tetravalent Dengue virus vaccine candidates in clinal trials including DENVax and TV003/TV005 (1-3).
References
1. Nanaware N, Banerjee A, Mullick Bagchi S, Bagchi P, Mukherjee A. Dengue Virus Infection: A Tale of Viral Exploitations and Host Responses. Viruses. 2021;13(10):1967. Published 2021 Sep 30. https://doi.org/10.3390/v13101967
2. Harapan H, Michie A, Sasmono RT, Imrie A. Dengue: A Minireview. Viruses. 2020;12(8):829. Published 2020 Jul 30. https://doi.org/10.3390/v12080829
3. Roy SK, Bhattacharjee S. Dengue virus: epidemiology, biology, and disease aetiology. Can J Microbiol. 2021;67(10):687-702. https://doi.org/10.1139/cjm-2020-0572
Alternate Names
polyprotein precursor
Product Documents for Recombinant Virus Dengue Virus 1 VLP Protein
Product Specific Notices for Recombinant Virus Dengue Virus 1 VLP Protein
This product is for research use only and is not approved for use in humans or in clinical diagnosis. This product is guaranteed for 1 year from date of receipt.
Loading...
Loading...
Loading...
Loading...