Recombinant Human TRIM59 GST (N-Term) Protein
Novus Biologicals, part of Bio-Techne | Catalog # H00286827-P01

Key Product Details
Source
Wheat germ
Tag
GST (N-Term)
Conjugate
Unconjugated
Applications
Western Blot, ELISA, Affinity Purification, Microarray
Product Specifications
Description
A recombinant protein with GST tag at N-terminal corresponding to the amino acids 1-403 of Human TRIM59
Source: Wheat Germ (in vitro)
Amino Acid Sequence: MHNFEEELTCPICYSIFEDPRVLPCSHTFCRNCLENILQASGNFYIWRPLRIPLKCPNCRSITEIAPTGIESLPVNFALRAIIEKYQQEDHPDIVTCPEHYRQPLNVYCLLDKKLVCGHCLTIGQHHGHPIDDLQSAYLKEKDTPQKLLEQLTDTHWTDLTHLIEKLKEQKSHSEKMIQGDKEAVLQYFKELNDTLEQKKKSFLTALCDVGNLINQEYTPQIERMKEIREQQLELMALTISLQEESPLKFLEKVDDVRQHVQILKQRPLPEVQPVEIYPRVSKILKEEWSRTEIGQIKNVLIPKMKISPKRMSCSWPGKDEKEVEFLKILNIVVVTLISVILMSILFFNQHIITFLSEITLIWFSEASLSVYQSLSNSLHKVKNILCHIFYLLKEFVWKIVSH
Purity
>80% by SDS-PAGE and Coomassie blue staining
Predicted Molecular Mass
73.5 kDa.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Activity
This protein was produced in an in vitro wheat germ expression system that should preserve correct conformational folding that is necessary for biological function. While it is possible that this protein could display some level of activity, the functionality of this protein has not been explicitly measured or validated.
Protein / Peptide Type
Recombinant Protein
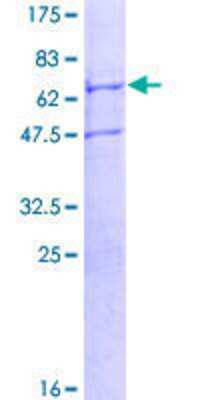
Scientific Data Images for Recombinant Human TRIM59 GST (N-Term) Protein
12.5% SDS-PAGE Stained with Coomassie Blue.
Formulation, Preparation and Storage
H00286827-P01
| Preparation Method | in vitro wheat germ expression system |
| Formulation | 50 mM Tris-HCl, 10 mM reduced Glutathione, pH 8.0 in the elution buffer. |
| Preservative | No Preservative |
| Concentration | Please see the vial label for concentration. If unlisted please contact technical services. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Store at -80C. Avoid freeze-thaw cycles. |
Background: TRIM59
1. Kimura, T., Mandell, M., & Deretic, V. (2016). Precision autophagy directed by receptor regulators - emerging examples within the TRIM family. Journal of Cell Science. https://doi.org/10.1242/jcs.163758
2. Wang, M., Chao, C., Luo, G., Wang, B., Zhan, X., Di, D., Qian, Y., & Zhang, X. (2019). Prognostic significance of TRIM59 for cancer patient survival: A systematic review and meta-analysis. Medicine, 98(48), e18024. https://doi.org/10.1097/MD.0000000000018024
3. Mandell, M. A., Saha, B., & Thompson, T. A. (2020). The Tripartite Nexus: Autophagy, Cancer, and Tripartite Motif-Containing Protein Family Members. Frontiers in Pharmacology. https://doi.org/10.3389/fphar.2020.00308
4. Chang, R., Xu, X., & Li, M. D. (2002). Molecular cloning, mapping and characterization of a novel mouse RING finger gene, Mrf1. Gene. https://doi.org/10.1016/S0378-1119(02)00603-0
Alternate Names
MGC129860, MGC129861, Mrf1, RING finger protein 104, RNF104MGC26631, TRIM57, tripartite motif containing 59, tripartite motif-containing 57, tripartite motif-containing 59, tripartite motif-containing protein 59, TSBF1MRF1, tumor suppressor TSBF1, Tumor suppressor TSBF-1
Gene Symbol
TRIM59
Additional TRIM59 Products
Product Documents for Recombinant Human TRIM59 GST (N-Term) Protein
Product Specific Notices for Recombinant Human TRIM59 GST (N-Term) Protein
This product is produced by and distributed for Abnova, a company based in Taiwan.
This product is for research use only and is not approved for use in humans or in clinical diagnosis. This product is guaranteed for 1 year from date of receipt.
Loading...
Loading...
Loading...
Loading...