Recombinant Human VEGF 165 GMP Protein, CF GMP Best Seller
R&D Systems, part of Bio-Techne | Catalog # BT-VEGF-GMP

Key Product Details
- VEGF Manufactured in Bio-Techne's new GMP facility
- Lot-to-lot consistency
- Stringent guidelines for patient safety
- Scalability necessary to support successful therapeutics
- Learn more about manufacturing in our new GMP facility
- Test it in your process! Request a sample of GMP VEGF
- A DMF is on file for this protein
Source
Accession #
Structure / Form
Conjugate
Applications
Product Specifications
Source
Ala27 - Arg191
Produced using non-animal reagents in an animal-free laboratory.Manufactured and tested under cGMP guidelines.
Purity
Endotoxin Level
N-terminal Sequence Analysis
Pro28-Met-Ala-Glu-Gly-Gly-Gly-Gln-Asn-His
Predicted Molecular Mass
SDS-PAGE
Activity
The ED50 for this effect is 1.50-12.0 ng/mL.
Host Cell Protein
Mycoplasma
Host Cell DNA
Scientific Data Images for Recombinant Human VEGF 165 GMP Protein, CF
Recombinant Human VEGF 165 GMP Protein Bioactivity.
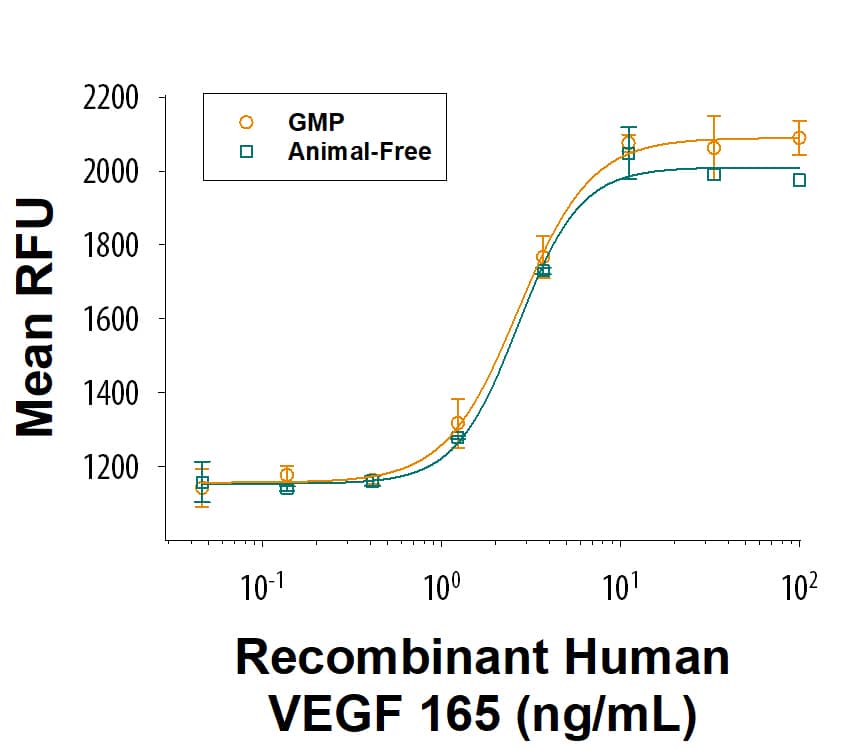
GMP-grade Recombinant Human VEGF 165 (Catalog # BT-VEGF-GMP) as measured in a cell proliferation assay using HUVEC human umbilical vein endothelial cells. The ED50 for this effect is 1.50-12.0 ng/mL. Three independent lots were tested for activity and plotted on the same graph to show lot-to-lot consistency of GMP VEGF 165.Equivalent Bioactivity of GMP and Animal-Free grades of Recombinant Human VEGF 165.
Equivalent bioactivity of GMP (Catalog # BT-VEGF-GMP) and Animal-Free (BT-VEGF-AFL) grades of Recombinant Human VEGF 165 as measured in a cell proliferation assay using HUVEC human umbilical vein endothelial cells (orange and green, respectively).Recombinant Human VEGF 165 GMP Protein SDS-PAGE
2 μg/lane of Recombinant Human VEGF 165 GMP Protein (Catalog # BT-VEGF-GMP) was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing bands at 19-21 kDa.Formulation, Preparation and Storage
BT-VEGF-GMP
| Formulation | Lyophilized from a 0.2 μm filtered solution in Sodium Acetate. |
| Reconstitution | Reconstitute at 500 μg/mL in sterile deionized water. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: VEGF
Vascular endothelial growth factor (VEGF or VEGF-A), also known as vascular permeability factor (VPF), is a potent mediator of both angiogenesis and vasculogenesis in the fetus and adult (1-3). It is a member of the PDGF family that is characterized by the presence of eight conserved cysteine residues and a cystine knot structure (4). Humans express alternately spliced isoforms of 121, 145, 165, 183, 189, and 206 amino acids (aa) in length (4). VEGF165 appears to be the most abundant and potent isoform, followed by VEGF121 and VEGF189 (3, 4). Isoforms other than VEGF121 contain basic heparin-binding regions and are not freely diffusible (4). Human VEGF165 shares 88% aa sequence identity with corresponding regions of mouse and rat, 96% with porcine, 95% with canine, and 93% with feline, equine and bovine VEGF, respectively. VEGF binds the type I transmembrane receptor tyrosine kinases VEGF R1 (also called Flt-1) and VEGF R2 (Flk-1/KDR) on endothelial cells (4). Although VEGF affinity is highest for binding to VEGF R1, VEGF R2 appears to be the primary mediator of VEGF angiogenic activity (3, 4). VEGF165 binds the semaphorin receptor, Neuropilin-1 and promotes complex formation with VEGF R2 (5). VEGF is required during embryogenesis to regulate the proliferation, migration, and survival of endothelial cells (3, 4). In adults, VEGF functions mainly in wound healing and the female reproductive cycle (3). Pathologically, it is involved in tumor angiogenesis and vascular leakage (6, 7). Circulating VEGF levels correlate with disease activity in autoimmune diseases such as rheumatoid arthritis, multiple sclerosis and systemic lupus erythematosus (8). VEGF is induced by hypoxia and cytokines such as IL-1, IL-6, IL-8, oncostatin M and TNF-alpha (3, 4, 9).
Due to its role in angiogenesis of blood vessels, tumor and stroma cells use VEGF to stimulate formation of blood vessels and the proliferation and survival of endothelial cells. Specific immunotherapies targeting the VEGF signaling pathway include the recombinant antibody against VEGF (Bevacizumab), antibodies targeting the main VEGF receptor (VEGFR2), and small molecule inhibitors against VEGF receptor tyrosine kinases (10). Immune checkpoint inhibitors are an important tool in cancer therapies as tumor cells can hijack immune checkpoint signals to evade detection by immune cells. In addition to stimulating the formation of tumor blood vessels, VEGF has immunosuppressive effects by acting on dendritic cells to block their antigen-presenting and T cell stimulatory functions. Targeting VEGF in combination with other immune checkpoint ligands or receptors may prove more effective in immunotherapy approaches to certain cancer types (11). Because of its role in the formation of blood vessels, VEGF is also an important factor in skeletal development where blood supply and vascularization are crucial. This has made VEGF an important molecule in regenerative studies for bone repair as sustained release of VEGF has been shown to improve the efficiency of bone regeneration (12).
In differentiation protocols for stems cells, VEGF is a commonly added growth factor for the transformation of induced pluripotent stem cells into hematopoietic progenitor cells used to make Natural Killer cells (13, 14). VEGF has also been used to transform intermediate mesoderm into kidney glomerular podocytes or stem cell-derived liver spheres (15, 16). VEGF may also be used in assistance of stem cell transplantations by supporting angiogenesis at sites of stem cell transplants or as a honing tool for adipose-derived mesenchymal stem cells or bone marrow stem cells to migrate to (17, 18).References
- Leung, D.W. et al. (1989) Science 246:1306.
- Keck, P.J. et al. (1989) Science 246:1309.
- Byrne, A.M. et al. (2005) J. Cell. Mol. Med. 9:777.
- Robinson, C.J. and S.E. Stringer (2001) J. Cell. Sci. 114:853.
- Pan, Q. et al. (2007) J. Biol. Chem. 282:24049.
- Weis, S.M. and D.A. Cheresh (2005) Nature 437:497.
- Thurston, G. (2002) J. Anat. 200:575.
- Carvalho, J.F. et al. (2007) J. Clin. Immunol. 27:246.
- Angelo, L.S. and R. Kurzrock (2007) Clin. Cancer Res. 13:2825.
- Apte, R.S. et al. (2019) Cell 176:1248.
- Sangro, B. et al. (2021) Nature 18:525.
- Hu, K. & Olsen, B.R. (2016) Bone 91:30.
- Zhou, Y. et al. (2022) Cancers 14:2266.
- Li, Y. et al. (2018) Cell Stem Cell. 23:181.
- Musah, S. et al. (2018) Nat. Protoc. 13:1662.
- Meseguer-Ripolles, J. et al. (2021) STAR Protoc. 2:100502.
- Hutchings, G. et al. (2020) Int. J. Mol. Sci. 21:3790.
- Zhang, W. et al. (2014) Eur. Cell. Mater. 27:1.
Long Name
Alternate Names
Entrez Gene IDs
Gene Symbol
UniProt
Additional VEGF Products
Product Documents for Recombinant Human VEGF 165 GMP Protein, CF
Manufacturing Specifications
GMP ProteinsR&D Systems, a Bio-Techne Brand's GMP proteins are produced according to relevant sections of the following documents: USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Eu. Ph. 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products.
R&D Systems' quality focus includes:
- Designed, manufactured and tested under an ISO 9001:2015 and ISO 13485:2016 certified quality system
- Documented and controlled manufacturing process
- Control of documentation and process changes by QA
- Personnel training programs
- Raw material inspection and vendor qualification/monitoring program
- Validated equipment, processes and test methods
- Equipment calibration and maintenance schedules using a Regulatory Asset Manager
- Facility/Utilities maintenance, contamination controls, safety and pest control programs
- Material review process for variances
- Robust product stability program following relevant ICH guidelines
- N-terminal amino acid analysis
- SDS-PAGE purity analysis
- Molecular weight analysis via mass spectrometry
- Endotoxin assessment per USP <85> and Ph. Eur. 2.6.14 guidelines
- Bioassay analysis
- Microbial testing per USP <71> and Ph. Eur. 2.6.1 guidelines
- Host cell protein assessment
- Host cell DNA assessment
- Mycoplasma assessment
Production records and facilities are available for examination by appropriate personnel on-site at R&D Systems in Minneapolis and St. Paul, Minnesota USA.
R&D Systems sells GMP grade products for preclinical or clinical ex vivo use. They are not for in vivo use. Please read the following End User Terms prior to using this product.
Animal-Free Manufacturing Conditions
Our dedicated controlled-access animal-free laboratories ensure that at no point in production are the products exposed to potential contamination by animal components or byproducts. Every stage of manufacturing is conducted in compliance with R&D Systems' stringent Standard Operating Procedures (SOPs). Production and purification procedures use equipment and media that are confirmed animal-free.
Production
- All molecular biology procedures use animal-free media and dedicated labware.
- Dedicated fermentors are utilized in committed animal-free areas.
- Protein purification columns are animal-free.
- Bulk proteins are filtered using animal-free filters.
- Purified proteins are stored in animal-free containers.
Product Specific Notices for Recombinant Human VEGF 165 GMP Protein, CF
Full terms and conditions of sale can be found online in the Protein Sciences Segment T&Cs at: Terms & Conditions.
For preclinical, or clinical ex vivo use