Human IL-33 Antibody
R&D Systems, part of Bio-Techne | Catalog # MAB3625

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Ser112-Thr270
Accession # O95760
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human IL-33 Antibody
Detection of IL-33 by Western Blot
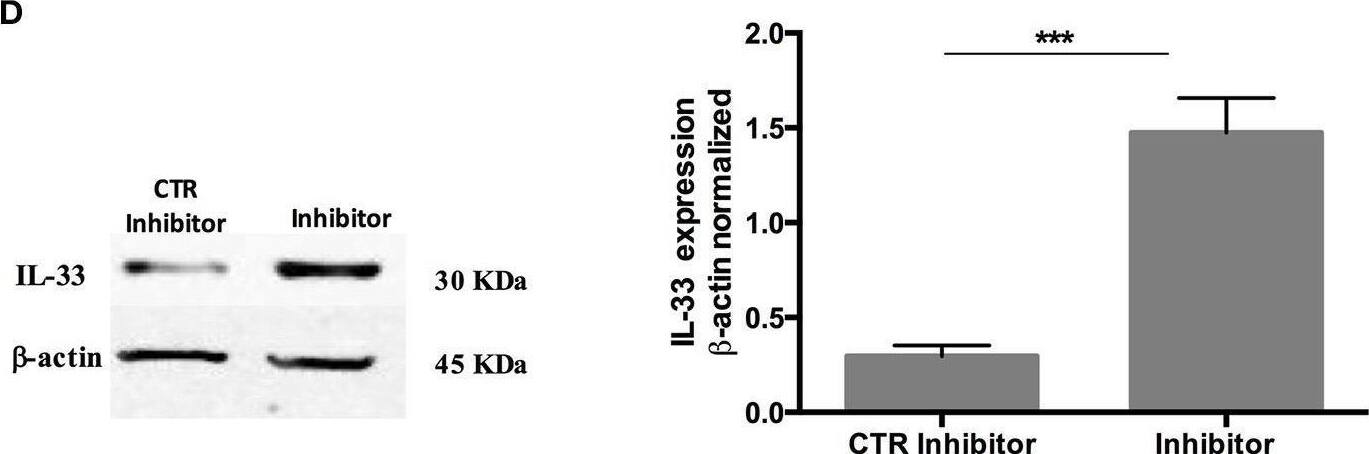
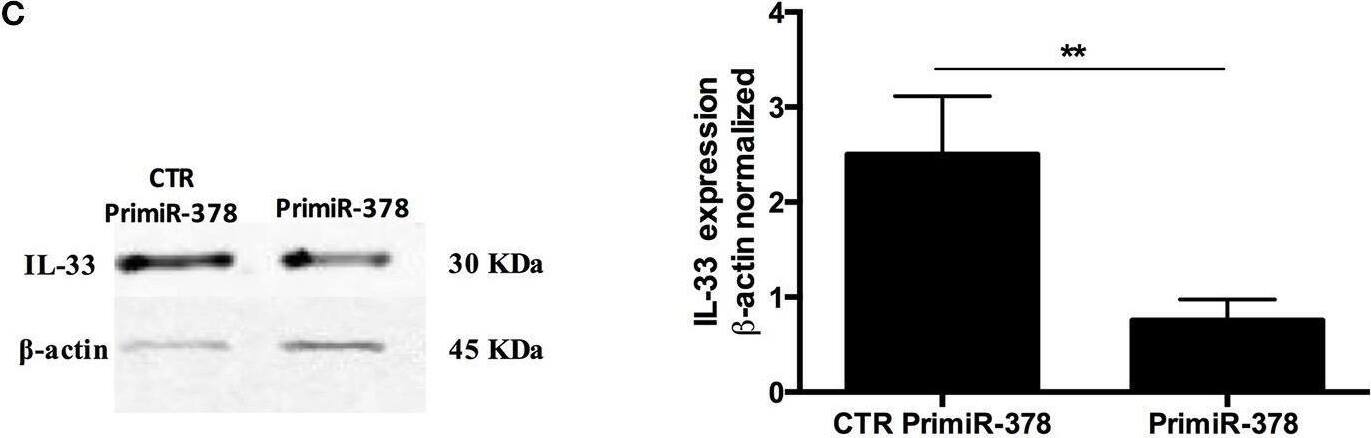
Over-expression and inhibition of miR-378a-3p modulate IL-33 expression. Stable expression of miR-378a-3p (Primir-378) or inhibition of miR-378a-3p (Inhibitor), and their respective controls (CTR PrimiR-378a, CTR Inhibitor) were obtained with Lentiviral vectors in HT-29 cells. (A) MiR-378a-3p Log2 fold change, and (B) IL-33 mRNA fold change relative to CTR cell lines by qPCR, (C) IL-33 protein and beta-actin by Western Blotting (WB) and bands quantification by densitometry analysis (Fiji Software) of Control Primir-378 and Primir-378 cell lines, and (D) Control Inhibitor and Inhibitor cell lines. Four experiments by duplicated were performed. Each cell line was compared with their respective control using Paired t-test. *P = < 0.05, **P = < 0.01, ***P = < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31824476), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
Over-expression and inhibition of miR-378a-3p modulate IL-33 expression. Stable expression of miR-378a-3p (Primir-378) or inhibition of miR-378a-3p (Inhibitor), and their respective controls (CTR PrimiR-378a, CTR Inhibitor) were obtained with Lentiviral vectors in HT-29 cells. (A) MiR-378a-3p Log2 fold change, and (B) IL-33 mRNA fold change relative to CTR cell lines by qPCR, (C) IL-33 protein and beta-actin by Western Blotting (WB) and bands quantification by densitometry analysis (Fiji Software) of Control Primir-378 and Primir-378 cell lines, and (D) Control Inhibitor and Inhibitor cell lines. Four experiments by duplicated were performed. Each cell line was compared with their respective control using Paired t-test. *P = < 0.05, **P = < 0.01, ***P = < 0.001. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31824476), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Western Blot
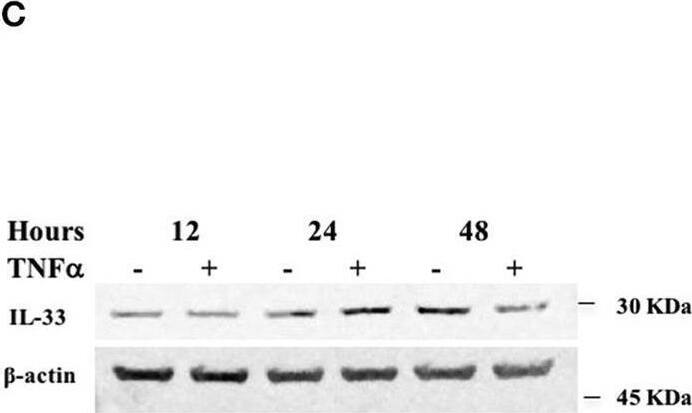
TNF alpha decreases miR-378a-3p and increases IL-33 expression in colonocytes. HT-29 cell line was stimulated with 10 ng/mL human recombinant TNF alpha (hr-TNF alpha) for 3, 6, and 24 h. Fold change relative to non-stimulated control of (A) MiR-378a-3p and (B) IL-33 mRNA. (C) IL-33 protein was measured in HT-29 cells stimulated with hr-TNF alpha 10 ng/mL 12, 24, and 48 h by WB, (D) Band quantification of IL-33 protein normalized to beta-actin. (E) Fold change relative to non-stimulated control of PPARGC1B mRNA and (F) IL-8 mRNA levels (measured as control of TNF alpha stimulus). (G) AGO1 and (H) AGO2 mRNA levels were assessed as control of miRNA-biogenesis machinery. Four experiments by duplicated were analyzed using Two-way ANOVA with Bonferroni post-test. *P < 0.05, **P = < 0.01. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31824476), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human IL-33 Antibody
Immunocytochemistry
Sample: Immersion fixed human peripheral blood mononuclear cells treated with treated with PMA and Ca2+ ionomycin
Western Blot
Sample: Recombinant Human IL-33 (Catalog # 3625-IL)
Reviewed Applications
Read 1 review rated 4 using MAB3625 in the following applications:
Formulation, Preparation, and Storage
Purification
Reconstitution
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Background: IL-33
IL-33, also known as NF-HEV and DVS 27, is a proinflammatory protein that may also regulate gene transcription (1-3). DVS 27 was identifed as a gene that is upregulated in vasospastic cerebral arteries (1). NF-HEV was described as a nuclear factor that is preferentially expressed in the endothelial cells of high endothelial venules relative to endothelial cells from other tissues (2). IL-33 was identified based on sequence and structural homology with IL-1 family cytokines (3). IL-33 is constitutively expressed in smooth muscle and airway epithelia. It is upregulated in arterial smooth muscle, dermal fibroblasts, and keratinocytes following IL-1 alpha or IL‑1 beta stimulation (1, 3). Similar to IL-1, IL-33 can be cleaved in vitro by caspase-1, generating an N-terminal fragment that is slightly shorter than the C-terminal fragment (3, 4). The N-terminal portion of full length 32 kDa IL-33 contains a predicted bipartite nuclear localization sequence and a homeodomain-like helix-turn-helix DNA binding domain. By immunofluorescence, full length IL-33 localizes to the nucleus in HUVECs and transfectants (2). The C-terminal 18 kDa fragment, corresponding to mature IL-33, binds and triggers signaling through mast cell IL-1 R4/ST2, a receptor involved in the augmentation of Th2 cell responses (3, 5-7). A ternary signaling complex is formed by the subsequent association of IL-33 and ST2 with IL-1R AcP (8). Stimulation of Th2 polarized lymphocytes with mature IL-33 in vitro induces IL-5 and IL-13 secretion (3). In vivo administration of mature IL-33 promotes increased production of IL-5, IL-13, IgE, and IgA, as well as splenomegaly and inflammatory infiltration of mucosal tissues (3). Full length and mature human IL-33 share 52‑58% aa sequence identity with mouse and rat IL-33. Human IL-33 shares less than 20% aa sequence identity with other IL-1 family proteins.
References
- Onda, H. et al. (1999) J. Cereb. Blood Flow Metab. 19:1279.
- Baekkevold, E.S. et al. (2003) Am. J. Pathol. 163:69.
- Schmitz, J. et al. (2005) Immunity 23:479.
- Black, R.A. et al. (1989) J. Biol. Chem. 264:5323.
- Xu, D. et al. (1998) J. Exp. Med. 187:787.
- Lohning, M. et al. (1998) Proc. Natl. Acad. Sci. USA 95:6930.
- Dinarello, C.A. (2005) Immunity 23:461.
- Chackerian, A.A. et al. (2007) J. Immunol. 179:2551.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional IL-33 Products
Product Documents for Human IL-33 Antibody
Product Specific Notices for Human IL-33 Antibody
For research use only