Human IgE Antibody (I27/mAb27) - Azide and BSA Free
Novus Biologicals, part of Bio-Techne | Catalog # DDX0290P-100

Key Product Details
Species Reactivity
Human
Applications
Western Blot, Sandwich ELISA, Block/Neutralize, Flow Cytometry, Functional
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # I27/mAb27
Format
Azide and BSA Free
Concentration
0.5 mg/ml
Product Specifications
Immunogen
Human IgE was used as immunogen.
Reactivity Notes
Human
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human IgE Antibody (I27/mAb27) - Azide and BSA Free
Flow Cytometry: Human IgE Antibody (I27/mAb27) [DDX0290P-100]
Flow Cytometry: Human IgE Antibody (I27/mAb27) [DDX0290P-100] - Fixation of mAb27 to IgEbound to FcεR2/CD23 on RPMI 8866 cells
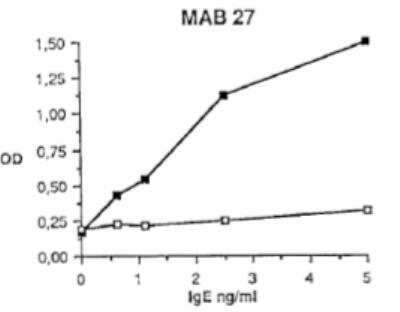
Sandwich ELISA: Human IgE Antibody (I27/mAb27) [DDX0290P-100] - Bindingof mAb27 to IgE (open) and heat denatured IgE (closed) in a sandwich ELISA
Applications for Human IgE Antibody (I27/mAb27) - Azide and BSA Free
Application
Recommended Usage
Flow Cytometry
1:10-1:1000
Sandwich ELISA
1:100-1:2000
Western Blot
1:100-1:2000
Formulation, Preparation, and Storage
Purification
Ion exchange chromatography
Formulation
Tris-NaCl (pH 8.0)
Format
Azide and BSA Free
Preservative
No Preservative
Concentration
0.5 mg/ml
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Store at -20C. Avoid freeze-thaw cycles.
Background: Human IgE
Alternate Names
immunoglobulin heavy constant epsilon
Additional Human IgE Products
Product Documents for Human IgE Antibody (I27/mAb27) - Azide and BSA Free
Product Specific Notices for Human IgE Antibody (I27/mAb27) - Azide and BSA Free
This product is manufactured by Eurobio Scientific (formerly Dendritics) and distributed by Novus Biologicals.
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Loading...
Loading...
Loading...
Loading...
Loading...
![Flow Cytometry: Human IgE Antibody (I27/mAb27) [DDX0290P-100] Flow Cytometry: Human IgE Antibody (I27/mAb27) [DDX0290P-100]](https://resources.bio-techne.com/images/products/Human-IgE-Antibody-I27-mAb27-Flow-Cytometry-DDX0290P-100-img0001.jpg)