Recombinant Human Noggin GMP Protein, CF GMP
R&D Systems, part of Bio-Techne | Catalog # 6057-GMP

Key Product Details
- A DMF is on file for this protein
Source
Accession #
Structure / Form
Conjugate
Applications
Product Specifications
Source
Gln28-Cys232
Manufactured and tested under current Good Manufacturing Practice (GMP) guidelines.
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted N-terminal sequence: Gln-His-Tyr-Leu-His-Ile-Arg-Pro-Ala-Pro
Predicted Molecular Mass
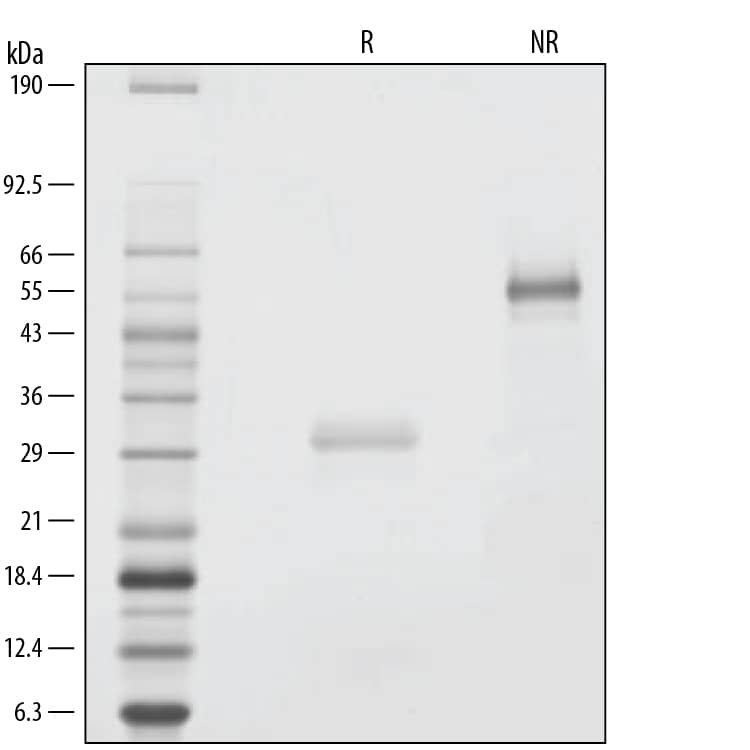
SDS-PAGE
Activity
The ED50 for this effect is 0.02-0.16 μg/mL in the presence of 50 ng/mL of Recombinant Human BMP‑4 (Catalog # 314-BP).
Host Cell Protein
Mycoplasma
Reviewed Applications
Read 1 review rated 4 using 6057-GMP in the following applications:
Scientific Data Images for Recombinant Human Noggin GMP Protein, CF
Recombinant Human Noggin GMP Protein Bioactivity
GMP-grade Recombinant Human Noggin (Catalog # 6057-GMP) inhibits BMP-4-induced alkaline phosphatase production in the ATDC5 mouse chondrogenic cell line. The ED50 for this effect is 0.04-0.2 µg/mL in the presence of 50 ng/mL of Recombinant Human BMP-4 (Catalog # 314-BP).Recombinant Human Noggin GMP Protein SDS-PAGE
1 μg/lane of GMP-grade Recombinant Human Noggin (Catalog # 6057-GMP) was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by silver staining, showing bands at 31 kDa and 57 kDa, respectively.Formulation, Preparation and Storage
6057-GMP
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 100 μg/mL in PBS. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: Noggin
Noggin is a secreted homodimeric glycoprotein that is an antagonist of bone morphogenetic proteins (BMPs) (1, 2). Human Noggin cDNA encodes a 232 amino acid (aa) precursor protein; cleavage of a 19 aa signal peptide generates the 213 aa mature protein which contains an N-terminal acidic region, a central basic heparin‑binding segment and a C-terminal cysteine-knot structure (2). Secreted Noggin probably remains close to the cell surface due to its binding of heparin‑containing proteoglycans (3). Noggin is very highly conserved among vertebrates, such that mature human Noggin shares 99%, 99%, 98%, 97% and 89% aa sequence identity with mouse, rat, bovine, equine and chicken Noggin, respectively. Noggin binds some BMPs such as BMP-4 with high affinity and others such as BMP-7 with lower affinity. It antagonizes BMP bioactivities by blocking epitopes on BMPs that are needed for binding to both type I and type II receptors (2, 4). During embryogenesis, Noggin antagonizes specific BMPs at defined times, for example, during neural tube, somite and cardiomyocyte growth and patterning (5-7). During skeletal development, Noggin prevents chondrocyte hyperplasia, thus allowing proper formation of joints (4). Mutations within the cysteine-knot region of human Noggin are linked to multiple types of skeletal dysplasias that result in apical joint fusions (8). Noggin is expressed in defined areas of the adult central nervous system and peripheral tissues such as lung, skeletal muscle and skin (1). During culture of human embryonic stem cells (hESC) or neural stem cells under certain conditions, addition of Noggin to antagonize BMP activity may allow stem cells to proliferate while maintaining their undifferentiated state, or alternatively, to differentiate into dopaminergic neurons (6, 9 - 13). Noggin also appears to maintain adult stem cell populations in-vivo, for example, maintaining neural stem cells within the hippocampus (13).
References
- Valenzuela, D.M. et al. (1995) J. Neurosci. 15:6077.
- Groppe, J. et al. (2002) Nature 420:636.
- Paine-Saunders, S et al. (2002) J. Biol. Chem. 277:2089.
- Brunet, L. J. et al. (1998) Science 280:1455.
- McMahon, J. A. et al. (1998) Genes Dev. 12:1438.
- Itsykson, P. et al. (2005) Mol. Cell. Neurosci. 30:24.
- Yuasa, S. et al. (2005) Nat. Biotechnol. 23:607.
- Gong, Y. et al. (1999) Nat. Genet. 21:302.
- Xu, R.-H. et al. (2005) Nat. Methods 2:185.
- Wang, G. et al. (2005) Biochem. Biophys. Res. Commun. 330:934.
- Chaturvedi, G. et al. (2009) Cell Prolif. 42:425.
- Chiba, S. et al. (2008) Stem Cells 26:2810.
- Bonaguidi, M.A. et al. (2008) J. Neurosci. 28:9194.
Alternate Names
Gene Symbol
UniProt
Additional Noggin Products
Product Documents for Recombinant Human Noggin GMP Protein, CF
Manufacturing Specifications
GMP ProteinsR&D Systems, a Bio-Techne Brand's GMP proteins are produced according to relevant sections of the following documents: USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Eu. Ph. 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products.
R&D Systems' quality focus includes:
- Designed, manufactured and tested under an ISO 9001:2015 and ISO 13485:2016 certified quality system
- Documented and controlled manufacturing process
- Control of documentation and process changes by QA
- Personnel training programs
- Raw material inspection and vendor qualification/monitoring program
- Validated equipment, processes and test methods
- Equipment calibration and maintenance schedules using a Regulatory Asset Manager
- Facility/Utilities maintenance, contamination controls, safety and pest control programs
- Material review process for variances
- Robust product stability program following relevant ICH guidelines
- N-terminal amino acid analysis
- SDS-PAGE purity analysis
- Molecular weight analysis via mass spectrometry
- Endotoxin assessment per USP <85> and Ph. Eur. 2.6.14 guidelines
- Bioassay analysis
- Microbial testing per USP <71> and Ph. Eur. 2.6.1 guidelines
- Host cell protein assessment
- Host cell DNA assessment
- Mycoplasma assessment
Production records and facilities are available for examination by appropriate personnel on-site at R&D Systems in Minneapolis and St. Paul, Minnesota USA.
R&D Systems sells GMP grade products for preclinical or clinical ex vivo use. They are not for in vivo use. Please read the following End User Terms prior to using this product.
Product Specific Notices for Recombinant Human Noggin GMP Protein, CF
Full terms and conditions of sale can be found online in the Protein Sciences Segment T&Cs at: Terms & Conditions.
For preclinical, or clinical ex vivo use