Human Alkaline Phosphatase/ALPL Antibody
R&D Systems, part of Bio-Techne | Catalog # MAB29092

Conjugate
Catalog #
Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human, Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Simple Western
Cited:
Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 928929
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Alkaline Phosphatase/ALPL
Leu18-Ser502
Accession # P05186
Leu18-Ser502
Accession # P05186
Specificity
Detects human Alkaline Phosphatase/ALPL in direct ELISA and Western Blots.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human Alkaline Phosphatase/ALPL Antibody
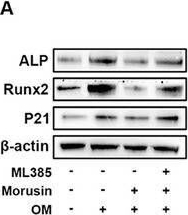
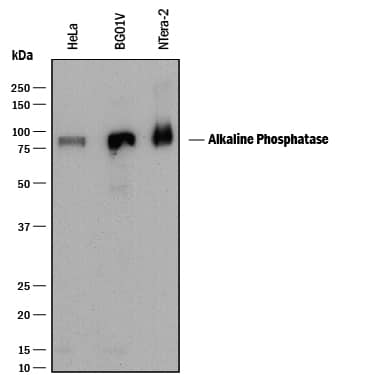
Detection of Human Alkaline Phosphatase/ALPL by Western Blot.
Western blot shows lysates of HeLa human cervical epithelial carcinoma cell line, BG01V human embryonic stem cells, and NTera-2 human testicular embryonic carcinoma cell line. PVDF membrane was probed with 0.25 µg/mL of Mouse Anti-Human Alkaline Phosphatase/ALPL Monoclonal Antibody (Catalog # MAB29092) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (HAF018). A specific band was detected for Alkaline Phosphatase/ALPL at approximately 80 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Alkaline Phosphatase/ALPL in Human Liver.
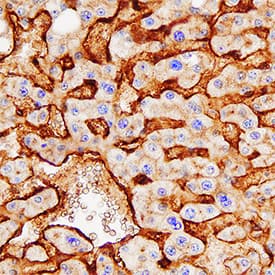
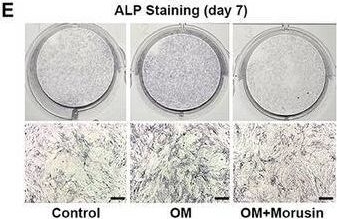
Alkaline Phosphatase/ALPL was detected in immersion fixed paraffin-embedded sections of human liver using Mouse Anti-Human Alkaline Phosphatase/ALPL Monoclonal Antibody (Catalog # MAB29092) at 0.3 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; CTS002) and counterstained with hematoxylin (blue). Specific staining was localized to bile canaliculi. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human Alkaline Phosphatase/ALPL by Simple WesternTM.
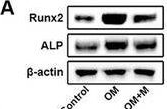
Simple Western lane view shows lysates of HeLa human cervical epithelial carcinoma cells, Saos-2 human osteosarcoma cells and BG01V human embryonic stem cells, loaded at 0.2 mg/mL. A specific band was detected for Alkaline Phosphatase/ALPL at approximately 115 kDa (as indicated) using 5 µg/mL of Mouse Anti-Human Alkaline Phosphatase/ALPL Monoclonal Antibody (Catalog # MAB29092) . This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Applications for Human Alkaline Phosphatase/ALPL Antibody
Application
Recommended Usage
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human liver
Sample: Immersion fixed paraffin-embedded sections of human liver
Simple Western
5 µg/mL
Sample: HeLa human cervical epithelial carcinoma cells, Saos-2 human osteosarcoma cells and BG01V human embryonic stem cells
Sample: HeLa human cervical epithelial carcinoma cells, Saos-2 human osteosarcoma cells and BG01V human embryonic stem cells
Western Blot
0.25 µg/mL
Sample: HeLa human cervical epithelial carcinoma cell line, BG01V human embryonic stem cells, and NTera‑2 human testicular embryonic carcinoma cell line
Sample: HeLa human cervical epithelial carcinoma cell line, BG01V human embryonic stem cells, and NTera‑2 human testicular embryonic carcinoma cell line
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Background: Alkaline Phosphatase/ALPL
Four distinct genes encode alkaline phosphatases (APs) in humans (1). The ALPL gene encodes the liver/bone/kidney isozyme, also known as the tissue-nonspecific AP (TNAP). In comparison, ALPI, ALPP and ALPPL2 encode intestinal, placental and placental-like or germ cell APs, respectively. The serum levels of human APs are useful tumor markers (2). There are many mutations in the ALPL gene, leading to different forms of hypophosphatasia, characterized by poorly mineralized cartilage and bones (3). The native ALPL is a glycosylated homodimer attached to the membrane through a GPI-anchor. The C-terminal pro peptide (residues 503‑524) is not present in the mature form.
References
- Le Du, M-H. and J.L. Millan (2002) J. Biol. Chem. 277:49808.
- Millan, J.L. and W.H. Fishman (1995) Crit. Rev. Clin. Lab. Sci. 32:1.
- Di Mauro, S. et al. (2002) J. Bone Miner. Res. 17:1383.
Long Name
Alkaline Phosphatase Liver
Alternate Names
Akp2, AP-TNAP, HOPS, TNAP, TNSALP
Gene Symbol
ALPL
UniProt
Additional Alkaline Phosphatase/ALPL Products
Product Documents for Human Alkaline Phosphatase/ALPL Antibody
Product Specific Notices for Human Alkaline Phosphatase/ALPL Antibody
For research use only
Loading...
Loading...
Loading...
Loading...
Loading...